-
PDF
- Split View
-
Views
-
Cite
Cite
Arthi Thirumalai, Fiona Yuen, John K Amory, Andrew N Hoofnagle, Ronald S Swerdloff, Peter Y Liu, Jill E Long, Diana L Blithe, Christina Wang, Stephanie T Page, Dimethandrolone Undecanoate, a Novel, Nonaromatizable Androgen, Increases P1NP in Healthy Men Over 28 Days, The Journal of Clinical Endocrinology & Metabolism, Volume 106, Issue 1, January 2021, Pages e171–e181, https://doi.org/10.1210/clinem/dgaa761
Close - Share Icon Share
Abstract
Dimethandrolone undecanoate (DMAU) is being developed as a male contraceptive. Daily oral administration of DMAU, a potent androgen that is not aromatized, markedly suppresses serum testosterone (T) and estradiol (E2) in healthy men. E2 deficiency can increase bone resorption in men.
This work aimed to assess changes in bone turnover markers with DMAU administration in a 28-day study.
A randomized, double-blind, placebo-controlled study was conducted.
This study took place at 2 academic medical centers.
Healthy men, age 18 to50 years (n = 81), participated.
Men received 0, 100, 200, or 400 mg of oral DMAU for 28 days. Serum C-terminal telopeptide of type I collagen (CTX; bone resorption marker) and procollagen type I amino-terminal propeptide (P1NP; bone formation marker) were measured on days 1 and 28.
Changes in bone turnover markers and serum hormones over the treatment period were measured.
On day 28, median serum T and E2 were markedly suppressed in all treatment groups vs placebo (P < .001 for both). Percentage change (%) in serum P1NP significantly differed across treatment groups (P = .007): Serum P1NP significantly increased in the 200 mg (5%, interquartile range [IQR] –7% to 27%) and 400 mg (22%, IQR –1% to 40%) groups relative to placebo (–8%, IQR –20% to 0%). Change (%) in serum CTX did not differ between groups (P = .09).
DMAU administration for 28 days to healthy men leads to marked suppression of serum T and E2, yet increases P1NP, a serum marker of bone formation. Longer-term studies of the potent androgen DMAU are warranted to determine its impact on bone health in men.
Gonadal steroids positively affect bone health in humans. Estrogens and testosterone (T) increase bone mineral density (BMD) during puberty by stimulating osteoblastic activity in females and males, respectively (1). During menopause, women lose considerable bone mass with the rapid decline in serum estradiol (E2) to prepubertal levels (2). Similarly, marked reductions in serum T concentrations in men, such as during androgen-deprivation therapy for prostate cancer, results in a sharp decline in BMD (3, 4). Moreover, even in men, estrogens likely play a major role in the maintenance of bone mass, independent of T (5).
Male hormonal contraception uses exogenous sex steroids to inhibit spermatogenesis (6). Exogenous steroids inhibit gonadotropin production, disrupting testicular steroidogenesis and sperm production while providing circulating androgens (and their metabolites such as E2) to nongonadal tissues. Although work to develop effective male hormonal contraceptives has been underway for decades, data regarding the impact of prototypic regimens on bone health are scant (7).
Male hormonal contraceptive regimens employ exogenous T—either alone or in combination with a progestin (8). Dosing of exogenous T to maintain serum T in the physiologic or supraphysiologic range is likely to maintain bone health in users since T is aromatized to maintain serum E2 concentrations (9, 10). Recently, novel androgens that have both androgenic and progestational activities are under investigation as potential oral male contraceptive agents, offering promise as a single-agent “male pill” (11, 12). One such agent is dimethandrolone undecanoate (DMAU), a prodrug that is converted to its active form, dimethandrolone (DMA), via cleavage of the undecanoate ester by endogenous esterases. DMA is greater than 4 times more potent than T at the androgen receptor (AR) (13). However, DMAU and DMA are not converted into active estrogenic compounds and are not aromatized in vivo (14). DMAU markedly suppresses serum T and E2 concentrations to castrate levels (11) in healthy men when administered for 28 days. This prompted us to explore whether DMAU may adversely affect bone formation and resorption in healthy men. In women, different progestins have been shown to have variable effects on bone health, with some causing bone loss by inhibition of gonadal steroid production (medroxyprogesterone acetate), whereas others show anabolic effects (norethindrone) (15). Such studies in men are scarce.
Typically, meaningful changes in BMD as a result of hormone treatment take many months to observe. However, bone turnover markers (BTMs) have been shown to change in response to anabolic therapy as early as 4 weeks (16), including following T enanthate injection, or T gel or sublingual T (17-19). Serum C-terminal telopeptide of type I collagen (CTX), a marker of bone resorption, and serum procollagen type I amino-terminal propeptide (P1NP), a marker of bone formation, have been proposed as the reference analytes for clinical and observational studies of bone health by the International Osteoporosis Foundation (20). Measurement of more than one BTM is generally preferred to corroborate the effects of any intervention on bone metabolism. However, most BTMs have specifications for sample collection and handling that can affect accuracy of results. Bone-specific alkaline phosphatase (BAP), also a marker of bone formation, is similar to P1NP in terms of sample collection and stability (21), but tends to show fewer alterations with treatment (22). Generally, bone resorption and formation are coupled processes; thus markers of bone turnover change in parallel (23). Longitudinal studies have demonstrated that simultaneous increases in serum CTX and P1NP are associated with accelerated bone loss (24) and increased fracture risk in men (25-27). However, with certain interventions, these 2 processes can become uncoupled, with a rise in bone formation markers (P1NP) but either no change (28) or decline (29) in resorption markers (CTX). The net effect of this uncoupling on BMD is unclear (28, 29).
We assessed the changes in serum CTX, P1NP, and BAP in samples from healthy men who participated in a 28-day study and received daily, oral DMAU (11). We hypothesized that despite very low serum T and E2 concentrations, BTMs would be maintained due to the potent androgenic actions exerted by circulating DMA, the active form of DMAU, at the AR (30).
Materials and Methods
Participants
This is a secondary analysis of samples from a previously published randomized, controlled trial of DMAU conducted at the University of Washington and The Lundquist Institute at Harbor–UCLA Medical Center (11), with Health Decisions as the coordinating and monitoring center. The institutional review boards for both participating clinical institutions and the single central review board (Chesapeake IRB) approved the study protocol. All participants provided written informed consent before initiation of any study procedures. Participants were healthy men, age 18 to 50 years, and equal numbers of individuals were enrolled at each site. Pertinent inclusion criteria included general good health and normal reproductive function, whereas exclusion criteria included significant medication use or chronic medical conditions. Details of these criteria, drug formulations, study design, and study procedures have been previously described in detail (11). Study participants took DMAU capsules daily for 28 consecutive days. Study visits included two 24-hour inpatient stays at the start and end of treatment to assess pharmacokinetics of the drug, and twice-weekly visits during the treatment period. The measurement of BTMs were not prespecified secondary end points of the original study; however, all included subjects consented to future analyses of stored serum specimens. Data on serum concentrations of hormones have been previously reported (11) and are included here by dose group for clarity.
Investigational drug
DMAU was manufactured by Evestra Inc or Ash Stevens Inc. Micronization was conducted by Micron Technologies Inc. Capsules containing 100 mg DMAU in powder blend or castor oil/benzyl benzoate, and corresponding placebo capsules, were manufactured, tested, and packaged under a cGMP (cyclic guanosine monophosphate) environment by Stanford Research International or QS Pharma.
Laboratory methods
Serum was stored frozen at –20°C. Measurement of serum hormone concentrations, chemistries, and hematologic parameters has been previously described (11). CTX and P1NP are known to be stable in samples stored at –20°C (31). We used serum from the –0.5-hour blood draw from 24-hour pharmacokinetics analyses performed on day 1 (drug start) and day 28 (or 29, end of treatment) of the parent study to measure serum CTX, P1NP, and BAP. This helped avoid variation in CTX measurements due to circadian rhythm or food intake, as per recommendations (32). We included samples from all participants in the efficacy evaluable population, defined as all treated participants who used at least 90% of the daily study drug and had taken their study drug within 1 day of the reported study visit. To eliminate the contribution of between-day variation in BTM measurements, all samples were run the same day in the Department of Laboratory Medicine of the University of Washington.
CTX was measured using the Serum Crosslaps EIA (enzyme immunoassay; Immunodiagnostic Systems, IDS-iSYS), a sandwich EIA with a lower limit of quantitation of 0.033 ng/mL and intra-assay and interassay coefficients of variation (CVs) of less than 6% and less than 10%, respectively. The Crosslaps EIA is an Food and Drug Administration–approved clinical diagnostic assay. P1NP was measured using an IDS-iSYS chemiluminometric assay with a lower limit of quantitation of 2 ng/mL and intra-assay and interassay CVs of less than 5% and less than 8.5%, respectively. Reference intervals in adult men for both these markers using these assays have been previously published (33, 34). BAP was quantified using an automated clinical immunoassay analyzer (Ostase, Beckman-Coulter DxI 800). The assay CV was 7.3% using the quality control material provided by the manufacturer, and the lower limit of quantification is 0.1 mcg/L.
Outcomes and statistical methods
The primary outcomes were the change in serum CTX and serum P1NP in each treatment group over the treatment period. Secondary outcomes consisted of exploring the relationships between BTM changes and changes in serum T, E2, and physiologic markers of androgenic activity (ie, hemoglobin).
The P1NP data were not normally distributed (even after log-transformation), so we employed nonparametric statistical tests for all analyses. Two different formulations (castor oil/benzyl benzoate and powder in capsule) of DMAU were tested in 3 different doses (100, 200, and 400 mg). For this analysis, we combined both formulations at any given dose level into one treatment group. While this is a retrospective analysis, based on the review of the means and SDs of previous studies that have measured changes in BTMs with sex-steroid manipulation in men, we estimated a greater than 90% power to see differences larger than 20% in serum CTX and P1NP between treatment groups at a significance level of .05.
We performed Kruskal-Wallis analysis of variance to assess for differences in CTX, P1NP, and BAP concentrations between all treatment groups on days 1 and 28/29. Wherever P was less than .05 for Kruskal-Wallis analysis of variance, we further performed pairwise comparisons using the Mann-Whitney U test with P less than .01 considered significant, adjusting for multiple comparisons. We calculated the change in CTX and P1NP as the difference in the values from day 28/29 to day 1 in each treatment group. To analyze whether the change or percentage change in CTX and P1NP concentrations from day 1 to day 28/29 within any given treatment group was significant, we performed the Wilcoxon signed-rank test, with P less than .05 considered significant.
In an exploratory analysis, we performed (univariate) Spearman correlation analyses looking at the relationship between change in serum P1NP and changes in serum T, serum E2, serum luteinizing hormone (LH), weight, serum high-density lipoprotein cholesterol (HDL-C), known androgenic variables (sex-hormone binding globulin [SHBG] and hematocrit [Hct]), dose of DMAU and serum drug concentrations (mean trough concentrations of serum DMA from days 2 to 28, maximum serum concentration of DMA on day 28, and area under the curve of serum DMA on day 28). We then performed reverse stepwise multivariate linear regression to explore the most significant contributor to the change in serum P1NP out of the following variables—age, baseline serum P1NP, change in serum T, change in serum E2, weight change, and dose of DMAU.
Results
Participants
A total of 202 research participants were screened, 100 were randomly assigned, 83 completed the study, and 82 men constituted the efficacy evaluable population (11). For 1 individuals, serum was not available on both visit days, leaving 81 participants included in this analysis. At baseline, participants in the DMAU 100 mg group were slightly older and serum CTX in that group was also slightly lower (Table 1). Otherwise, participants across the groups were comparable.
Baseline characteristics of participants by treatment group (median, interquartile range)
| Variable . | Placebo . | 100 mg . | 200 mg . | 400 mg . | P . |
|---|---|---|---|---|---|
| No. of participants | 23 | 13 | 26 | 19 | |
| Age, y | 26 (24-33) | 35 (31-42) | 28 (24-32) | 31 (26-37) | .02a,b |
| Weight, kg | 85 (74.1-90.8) | 89.7 (78.8-102.4) | 81.4 (75.2-90.9) | 78.4 (67.5-87.6) | .13 |
| BMI, kg/m2 | 25 (23-27) | 30 (26-31) | 25 (23.3-27.8) | 25 (2328) | .05 |
| Serum T, ng/dL | 477 (415-571) | 390 (316-449) | 485 (366-565) | 492 (442581) | .08 |
| Serum E2, pg/mL | 20.8 (8.5-24.8) | 24.1 (14.1-25.4) | 18.5 (10.4-28.1) | 15.4 (12.521.1) | .75 |
| Serum CTX, ng/mL | 0.27 (0.19-0.32) | 0.16 (0.13-0.22) | 0.24 (0.18-0.3) | 0.18 (0.14-0.24) | .03a,c |
| Serum P1NP, ng/mL | 67.7 (54.5-82.9) | 54 (35.3-67.8) | 57.9 (44.6-68.9) | 64 (55.8-83.2) | .14 |
| Serum BAP, mcg/L | 6.9 (5.8-8.5) | 6.4 (5.9-6.7) | 6.6 (6.3-8.3) | 6.7 (5.6-7.3) | .44 |
| Variable . | Placebo . | 100 mg . | 200 mg . | 400 mg . | P . |
|---|---|---|---|---|---|
| No. of participants | 23 | 13 | 26 | 19 | |
| Age, y | 26 (24-33) | 35 (31-42) | 28 (24-32) | 31 (26-37) | .02a,b |
| Weight, kg | 85 (74.1-90.8) | 89.7 (78.8-102.4) | 81.4 (75.2-90.9) | 78.4 (67.5-87.6) | .13 |
| BMI, kg/m2 | 25 (23-27) | 30 (26-31) | 25 (23.3-27.8) | 25 (2328) | .05 |
| Serum T, ng/dL | 477 (415-571) | 390 (316-449) | 485 (366-565) | 492 (442581) | .08 |
| Serum E2, pg/mL | 20.8 (8.5-24.8) | 24.1 (14.1-25.4) | 18.5 (10.4-28.1) | 15.4 (12.521.1) | .75 |
| Serum CTX, ng/mL | 0.27 (0.19-0.32) | 0.16 (0.13-0.22) | 0.24 (0.18-0.3) | 0.18 (0.14-0.24) | .03a,c |
| Serum P1NP, ng/mL | 67.7 (54.5-82.9) | 54 (35.3-67.8) | 57.9 (44.6-68.9) | 64 (55.8-83.2) | .14 |
| Serum BAP, mcg/L | 6.9 (5.8-8.5) | 6.4 (5.9-6.7) | 6.6 (6.3-8.3) | 6.7 (5.6-7.3) | .44 |
P values of Kruskal-Wallis analysis of variance across groups.
Abbreviations: BAP, bone-specific alkaline phosphatase; BMI, body mass index; CTX, C-terminal telopeptide of type I collagen; E2, estradiol; P1NP, procollagen type I amino-terminal propeptide; T, testosterone.
aP less than .05 was significant.
bSignificant pairwise comparisons were placebo vs 100 mg and 100 mg vs 200 mg.
cSignificant pairwise comparison was 100 mg vs 200 mg.
Baseline characteristics of participants by treatment group (median, interquartile range)
| Variable . | Placebo . | 100 mg . | 200 mg . | 400 mg . | P . |
|---|---|---|---|---|---|
| No. of participants | 23 | 13 | 26 | 19 | |
| Age, y | 26 (24-33) | 35 (31-42) | 28 (24-32) | 31 (26-37) | .02a,b |
| Weight, kg | 85 (74.1-90.8) | 89.7 (78.8-102.4) | 81.4 (75.2-90.9) | 78.4 (67.5-87.6) | .13 |
| BMI, kg/m2 | 25 (23-27) | 30 (26-31) | 25 (23.3-27.8) | 25 (2328) | .05 |
| Serum T, ng/dL | 477 (415-571) | 390 (316-449) | 485 (366-565) | 492 (442581) | .08 |
| Serum E2, pg/mL | 20.8 (8.5-24.8) | 24.1 (14.1-25.4) | 18.5 (10.4-28.1) | 15.4 (12.521.1) | .75 |
| Serum CTX, ng/mL | 0.27 (0.19-0.32) | 0.16 (0.13-0.22) | 0.24 (0.18-0.3) | 0.18 (0.14-0.24) | .03a,c |
| Serum P1NP, ng/mL | 67.7 (54.5-82.9) | 54 (35.3-67.8) | 57.9 (44.6-68.9) | 64 (55.8-83.2) | .14 |
| Serum BAP, mcg/L | 6.9 (5.8-8.5) | 6.4 (5.9-6.7) | 6.6 (6.3-8.3) | 6.7 (5.6-7.3) | .44 |
| Variable . | Placebo . | 100 mg . | 200 mg . | 400 mg . | P . |
|---|---|---|---|---|---|
| No. of participants | 23 | 13 | 26 | 19 | |
| Age, y | 26 (24-33) | 35 (31-42) | 28 (24-32) | 31 (26-37) | .02a,b |
| Weight, kg | 85 (74.1-90.8) | 89.7 (78.8-102.4) | 81.4 (75.2-90.9) | 78.4 (67.5-87.6) | .13 |
| BMI, kg/m2 | 25 (23-27) | 30 (26-31) | 25 (23.3-27.8) | 25 (2328) | .05 |
| Serum T, ng/dL | 477 (415-571) | 390 (316-449) | 485 (366-565) | 492 (442581) | .08 |
| Serum E2, pg/mL | 20.8 (8.5-24.8) | 24.1 (14.1-25.4) | 18.5 (10.4-28.1) | 15.4 (12.521.1) | .75 |
| Serum CTX, ng/mL | 0.27 (0.19-0.32) | 0.16 (0.13-0.22) | 0.24 (0.18-0.3) | 0.18 (0.14-0.24) | .03a,c |
| Serum P1NP, ng/mL | 67.7 (54.5-82.9) | 54 (35.3-67.8) | 57.9 (44.6-68.9) | 64 (55.8-83.2) | .14 |
| Serum BAP, mcg/L | 6.9 (5.8-8.5) | 6.4 (5.9-6.7) | 6.6 (6.3-8.3) | 6.7 (5.6-7.3) | .44 |
P values of Kruskal-Wallis analysis of variance across groups.
Abbreviations: BAP, bone-specific alkaline phosphatase; BMI, body mass index; CTX, C-terminal telopeptide of type I collagen; E2, estradiol; P1NP, procollagen type I amino-terminal propeptide; T, testosterone.
aP less than .05 was significant.
bSignificant pairwise comparisons were placebo vs 100 mg and 100 mg vs 200 mg.
cSignificant pairwise comparison was 100 mg vs 200 mg.
Serum hormones and androgenic end points
Serum T, E2, and SHBG were all markedly suppressed in active treatment groups compared to placebo in a dose-responsive manner (Table 2). Furthermore, a dose-responsive increase in weight, reductions in HDL-C, and increase in Hct in all DMAU groups were noted (see Table 2).
Changes in variables by treatment group from baseline to day 28 (median, interquartile range)
| Variable . | . | Placebo . | 100 mg . | 200 mg . | 400 mg . | P . |
|---|---|---|---|---|---|---|
| Serum T, ng/dL | Day 28 | 463 (387 to 527) | 26 (10 to 164) | 32 (15 to 102) | 11 (8 to 17) | < .001a |
| Change | –27 (–77 to 20) | –274 (–380 to –226) | –389 (–386 to –308) | –478 (–539 to –422) | < .001a | |
| Serum E2, pg/mL | Day 28 | 20.3 (16.4 to 23.7) | 8 (5.3 to 16.2) | 4.5 (3.1 to 8) | 4.2 (0.2 to 5.1) | < .001a |
| Change | 0.4 (–5.9 to 9.9) | –9.9 (–14.8 to –0.9) | –10.5 (–17.7 to –5.6) | –14.4 (–19.3 to –9.3) | < .001a | |
| Weight, kg | Day 28 | 85.3 (72.9 to 91.6) | 92 (81 to 103.5) | 83.1 (76.3 to 94.1) | 81.9 (71 to 90.8) | .17 |
| Change | 0.4 (–0.7 to 1.4) | 1.5 (1 to 1.2) | 1.8 (1.2 to 3.1) | 3.3 (1.5 to 3.9) | < .001a | |
| Hct, % | Day 28 | 40 (38.9 to 42.3) | 41.2 (40 to 43.1) | 44 (43 to 45.4) | 43.2 (40.5 to 44) | .008a |
| Change | –1 (–1.9 to –0.5) | 1 (0 to 2.7) | 1 (0 to 2.1) | 1 (–0.4 to 2.6) | < .001a | |
| HDL-C, mg/dL | Day 28 | 50 (41.5 to 53) | 33.5 (30.8 to 42.5) | 36 (32 to 45) | 34 (27.5 to 45.5) | .003a |
| Change | –1 (–5.5 to 1.8) | –7 (–11.3 to –4.8) | –11 (–16 to –8) | –13 (–20.5 to –9) | < .001a | |
| SHBG, nmol/L | Day 28 | 27.3 (18.3 to 35.3) | 12.6 (9.6 to 17.3) | 11.2 (8.6 to 13.3) | 9.2 (7.2 to 11.6) | < .001a |
| Change | –1.8 (–9.6 to 1.4) | –12.3 (–12.6 to –3.7) | –15.7 (–19.8 to –10.1) | –17.9 (–30.8 to –11.3) | < .001a | |
| Serum CTX, ng/mL | Day 28 | 0.18 (0.14 to 0.36) | 0.16 (0.15 to 0.24) | 0.21 (0.16 to 0.35) | 0.14 (0.08 to 0.18) | .006a |
| Change | –0.04 (–0.12 to 0.04) | 0 (–0.02 to 0.05) | –0.01 (–0.06 to 0.08) | –0.05 (–0.09 to 0.01) | .19 | |
| % Change | –11 (–40 to 24) | –4 (–11 to 43) | –2 (–26 to 44) | –24 (–55 to 11) | .09 | |
| Serum P1NP, ng/mL | Day 28 | 68.6 (40 to 80.1) | 48.8 (43.3 to 77.9) | 66.6 (55.2 to 89.9) | 75.3 (60.6 to 101.7) | .07 |
| Change | –6.6 (–11 to 0.3) | –0.5 (–6.3 to 10.1) | 2.8 (–4.6 to 17.9) | 12.1 (–0.6 to 32.1) | .005a | |
| % Change | –8 (–20 to 0) | –1 (–11 to 15) | 5 (–7 to 27) | 22 (–1 to 40) | .007a | |
| Serum BAP, mcg/L | Day 28 | 6.3 (5.6 to 7.5) | 6.0 (5.3 to 6.6) | 6.8 (6 to 8.4) | 6.5 (5.6 to 7.3) | .18 |
| Change | –0.6 (–1.2 to 0.1) | –0.2 (–0.5 to –0.1) | –0.3 (–0.7 to 0.3) | –0.2 (–0.5 to 0.4) | .31 | |
| % Change | –8.9 (–17.7 to 1.9) | –4.6 (–9.9 to –1.8) | –4.1 (–10.4 to 5.1) | –4.1 (–7.8 to 6.3) | .27 |
| Variable . | . | Placebo . | 100 mg . | 200 mg . | 400 mg . | P . |
|---|---|---|---|---|---|---|
| Serum T, ng/dL | Day 28 | 463 (387 to 527) | 26 (10 to 164) | 32 (15 to 102) | 11 (8 to 17) | < .001a |
| Change | –27 (–77 to 20) | –274 (–380 to –226) | –389 (–386 to –308) | –478 (–539 to –422) | < .001a | |
| Serum E2, pg/mL | Day 28 | 20.3 (16.4 to 23.7) | 8 (5.3 to 16.2) | 4.5 (3.1 to 8) | 4.2 (0.2 to 5.1) | < .001a |
| Change | 0.4 (–5.9 to 9.9) | –9.9 (–14.8 to –0.9) | –10.5 (–17.7 to –5.6) | –14.4 (–19.3 to –9.3) | < .001a | |
| Weight, kg | Day 28 | 85.3 (72.9 to 91.6) | 92 (81 to 103.5) | 83.1 (76.3 to 94.1) | 81.9 (71 to 90.8) | .17 |
| Change | 0.4 (–0.7 to 1.4) | 1.5 (1 to 1.2) | 1.8 (1.2 to 3.1) | 3.3 (1.5 to 3.9) | < .001a | |
| Hct, % | Day 28 | 40 (38.9 to 42.3) | 41.2 (40 to 43.1) | 44 (43 to 45.4) | 43.2 (40.5 to 44) | .008a |
| Change | –1 (–1.9 to –0.5) | 1 (0 to 2.7) | 1 (0 to 2.1) | 1 (–0.4 to 2.6) | < .001a | |
| HDL-C, mg/dL | Day 28 | 50 (41.5 to 53) | 33.5 (30.8 to 42.5) | 36 (32 to 45) | 34 (27.5 to 45.5) | .003a |
| Change | –1 (–5.5 to 1.8) | –7 (–11.3 to –4.8) | –11 (–16 to –8) | –13 (–20.5 to –9) | < .001a | |
| SHBG, nmol/L | Day 28 | 27.3 (18.3 to 35.3) | 12.6 (9.6 to 17.3) | 11.2 (8.6 to 13.3) | 9.2 (7.2 to 11.6) | < .001a |
| Change | –1.8 (–9.6 to 1.4) | –12.3 (–12.6 to –3.7) | –15.7 (–19.8 to –10.1) | –17.9 (–30.8 to –11.3) | < .001a | |
| Serum CTX, ng/mL | Day 28 | 0.18 (0.14 to 0.36) | 0.16 (0.15 to 0.24) | 0.21 (0.16 to 0.35) | 0.14 (0.08 to 0.18) | .006a |
| Change | –0.04 (–0.12 to 0.04) | 0 (–0.02 to 0.05) | –0.01 (–0.06 to 0.08) | –0.05 (–0.09 to 0.01) | .19 | |
| % Change | –11 (–40 to 24) | –4 (–11 to 43) | –2 (–26 to 44) | –24 (–55 to 11) | .09 | |
| Serum P1NP, ng/mL | Day 28 | 68.6 (40 to 80.1) | 48.8 (43.3 to 77.9) | 66.6 (55.2 to 89.9) | 75.3 (60.6 to 101.7) | .07 |
| Change | –6.6 (–11 to 0.3) | –0.5 (–6.3 to 10.1) | 2.8 (–4.6 to 17.9) | 12.1 (–0.6 to 32.1) | .005a | |
| % Change | –8 (–20 to 0) | –1 (–11 to 15) | 5 (–7 to 27) | 22 (–1 to 40) | .007a | |
| Serum BAP, mcg/L | Day 28 | 6.3 (5.6 to 7.5) | 6.0 (5.3 to 6.6) | 6.8 (6 to 8.4) | 6.5 (5.6 to 7.3) | .18 |
| Change | –0.6 (–1.2 to 0.1) | –0.2 (–0.5 to –0.1) | –0.3 (–0.7 to 0.3) | –0.2 (–0.5 to 0.4) | .31 | |
| % Change | –8.9 (–17.7 to 1.9) | –4.6 (–9.9 to –1.8) | –4.1 (–10.4 to 5.1) | –4.1 (–7.8 to 6.3) | .27 |
Change calculated as day 28 value minus day 1 value.
Abbreviations: BAP, bone-specific alkaline phosphatase; CTX, C-terminal telopeptide of type I collagen; E2, estradiol; Hct, hematocrit; HDL-C, high-density lipoprotein cholesterol; P1NP, procollagen type I amino-terminal propeptide; SHBG, sex-hormone binding globulin; T, testosterone.
aP less than .05 on Kruskal-Wallis analysis of variance.
Changes in variables by treatment group from baseline to day 28 (median, interquartile range)
| Variable . | . | Placebo . | 100 mg . | 200 mg . | 400 mg . | P . |
|---|---|---|---|---|---|---|
| Serum T, ng/dL | Day 28 | 463 (387 to 527) | 26 (10 to 164) | 32 (15 to 102) | 11 (8 to 17) | < .001a |
| Change | –27 (–77 to 20) | –274 (–380 to –226) | –389 (–386 to –308) | –478 (–539 to –422) | < .001a | |
| Serum E2, pg/mL | Day 28 | 20.3 (16.4 to 23.7) | 8 (5.3 to 16.2) | 4.5 (3.1 to 8) | 4.2 (0.2 to 5.1) | < .001a |
| Change | 0.4 (–5.9 to 9.9) | –9.9 (–14.8 to –0.9) | –10.5 (–17.7 to –5.6) | –14.4 (–19.3 to –9.3) | < .001a | |
| Weight, kg | Day 28 | 85.3 (72.9 to 91.6) | 92 (81 to 103.5) | 83.1 (76.3 to 94.1) | 81.9 (71 to 90.8) | .17 |
| Change | 0.4 (–0.7 to 1.4) | 1.5 (1 to 1.2) | 1.8 (1.2 to 3.1) | 3.3 (1.5 to 3.9) | < .001a | |
| Hct, % | Day 28 | 40 (38.9 to 42.3) | 41.2 (40 to 43.1) | 44 (43 to 45.4) | 43.2 (40.5 to 44) | .008a |
| Change | –1 (–1.9 to –0.5) | 1 (0 to 2.7) | 1 (0 to 2.1) | 1 (–0.4 to 2.6) | < .001a | |
| HDL-C, mg/dL | Day 28 | 50 (41.5 to 53) | 33.5 (30.8 to 42.5) | 36 (32 to 45) | 34 (27.5 to 45.5) | .003a |
| Change | –1 (–5.5 to 1.8) | –7 (–11.3 to –4.8) | –11 (–16 to –8) | –13 (–20.5 to –9) | < .001a | |
| SHBG, nmol/L | Day 28 | 27.3 (18.3 to 35.3) | 12.6 (9.6 to 17.3) | 11.2 (8.6 to 13.3) | 9.2 (7.2 to 11.6) | < .001a |
| Change | –1.8 (–9.6 to 1.4) | –12.3 (–12.6 to –3.7) | –15.7 (–19.8 to –10.1) | –17.9 (–30.8 to –11.3) | < .001a | |
| Serum CTX, ng/mL | Day 28 | 0.18 (0.14 to 0.36) | 0.16 (0.15 to 0.24) | 0.21 (0.16 to 0.35) | 0.14 (0.08 to 0.18) | .006a |
| Change | –0.04 (–0.12 to 0.04) | 0 (–0.02 to 0.05) | –0.01 (–0.06 to 0.08) | –0.05 (–0.09 to 0.01) | .19 | |
| % Change | –11 (–40 to 24) | –4 (–11 to 43) | –2 (–26 to 44) | –24 (–55 to 11) | .09 | |
| Serum P1NP, ng/mL | Day 28 | 68.6 (40 to 80.1) | 48.8 (43.3 to 77.9) | 66.6 (55.2 to 89.9) | 75.3 (60.6 to 101.7) | .07 |
| Change | –6.6 (–11 to 0.3) | –0.5 (–6.3 to 10.1) | 2.8 (–4.6 to 17.9) | 12.1 (–0.6 to 32.1) | .005a | |
| % Change | –8 (–20 to 0) | –1 (–11 to 15) | 5 (–7 to 27) | 22 (–1 to 40) | .007a | |
| Serum BAP, mcg/L | Day 28 | 6.3 (5.6 to 7.5) | 6.0 (5.3 to 6.6) | 6.8 (6 to 8.4) | 6.5 (5.6 to 7.3) | .18 |
| Change | –0.6 (–1.2 to 0.1) | –0.2 (–0.5 to –0.1) | –0.3 (–0.7 to 0.3) | –0.2 (–0.5 to 0.4) | .31 | |
| % Change | –8.9 (–17.7 to 1.9) | –4.6 (–9.9 to –1.8) | –4.1 (–10.4 to 5.1) | –4.1 (–7.8 to 6.3) | .27 |
| Variable . | . | Placebo . | 100 mg . | 200 mg . | 400 mg . | P . |
|---|---|---|---|---|---|---|
| Serum T, ng/dL | Day 28 | 463 (387 to 527) | 26 (10 to 164) | 32 (15 to 102) | 11 (8 to 17) | < .001a |
| Change | –27 (–77 to 20) | –274 (–380 to –226) | –389 (–386 to –308) | –478 (–539 to –422) | < .001a | |
| Serum E2, pg/mL | Day 28 | 20.3 (16.4 to 23.7) | 8 (5.3 to 16.2) | 4.5 (3.1 to 8) | 4.2 (0.2 to 5.1) | < .001a |
| Change | 0.4 (–5.9 to 9.9) | –9.9 (–14.8 to –0.9) | –10.5 (–17.7 to –5.6) | –14.4 (–19.3 to –9.3) | < .001a | |
| Weight, kg | Day 28 | 85.3 (72.9 to 91.6) | 92 (81 to 103.5) | 83.1 (76.3 to 94.1) | 81.9 (71 to 90.8) | .17 |
| Change | 0.4 (–0.7 to 1.4) | 1.5 (1 to 1.2) | 1.8 (1.2 to 3.1) | 3.3 (1.5 to 3.9) | < .001a | |
| Hct, % | Day 28 | 40 (38.9 to 42.3) | 41.2 (40 to 43.1) | 44 (43 to 45.4) | 43.2 (40.5 to 44) | .008a |
| Change | –1 (–1.9 to –0.5) | 1 (0 to 2.7) | 1 (0 to 2.1) | 1 (–0.4 to 2.6) | < .001a | |
| HDL-C, mg/dL | Day 28 | 50 (41.5 to 53) | 33.5 (30.8 to 42.5) | 36 (32 to 45) | 34 (27.5 to 45.5) | .003a |
| Change | –1 (–5.5 to 1.8) | –7 (–11.3 to –4.8) | –11 (–16 to –8) | –13 (–20.5 to –9) | < .001a | |
| SHBG, nmol/L | Day 28 | 27.3 (18.3 to 35.3) | 12.6 (9.6 to 17.3) | 11.2 (8.6 to 13.3) | 9.2 (7.2 to 11.6) | < .001a |
| Change | –1.8 (–9.6 to 1.4) | –12.3 (–12.6 to –3.7) | –15.7 (–19.8 to –10.1) | –17.9 (–30.8 to –11.3) | < .001a | |
| Serum CTX, ng/mL | Day 28 | 0.18 (0.14 to 0.36) | 0.16 (0.15 to 0.24) | 0.21 (0.16 to 0.35) | 0.14 (0.08 to 0.18) | .006a |
| Change | –0.04 (–0.12 to 0.04) | 0 (–0.02 to 0.05) | –0.01 (–0.06 to 0.08) | –0.05 (–0.09 to 0.01) | .19 | |
| % Change | –11 (–40 to 24) | –4 (–11 to 43) | –2 (–26 to 44) | –24 (–55 to 11) | .09 | |
| Serum P1NP, ng/mL | Day 28 | 68.6 (40 to 80.1) | 48.8 (43.3 to 77.9) | 66.6 (55.2 to 89.9) | 75.3 (60.6 to 101.7) | .07 |
| Change | –6.6 (–11 to 0.3) | –0.5 (–6.3 to 10.1) | 2.8 (–4.6 to 17.9) | 12.1 (–0.6 to 32.1) | .005a | |
| % Change | –8 (–20 to 0) | –1 (–11 to 15) | 5 (–7 to 27) | 22 (–1 to 40) | .007a | |
| Serum BAP, mcg/L | Day 28 | 6.3 (5.6 to 7.5) | 6.0 (5.3 to 6.6) | 6.8 (6 to 8.4) | 6.5 (5.6 to 7.3) | .18 |
| Change | –0.6 (–1.2 to 0.1) | –0.2 (–0.5 to –0.1) | –0.3 (–0.7 to 0.3) | –0.2 (–0.5 to 0.4) | .31 | |
| % Change | –8.9 (–17.7 to 1.9) | –4.6 (–9.9 to –1.8) | –4.1 (–10.4 to 5.1) | –4.1 (–7.8 to 6.3) | .27 |
Change calculated as day 28 value minus day 1 value.
Abbreviations: BAP, bone-specific alkaline phosphatase; CTX, C-terminal telopeptide of type I collagen; E2, estradiol; Hct, hematocrit; HDL-C, high-density lipoprotein cholesterol; P1NP, procollagen type I amino-terminal propeptide; SHBG, sex-hormone binding globulin; T, testosterone.
aP less than .05 on Kruskal-Wallis analysis of variance.
Bone turnover markers
Overall serum P1NP increased across treatment groups (P = .007) (Fig. 1A). Both the DMAU 200 mg (P = .006) and the DMAU 400 mg (P = .002) groups had significant increases in serum P1NP relative to placebo. Within the groups, during the treatment period, serum P1NP increased significantly only in the DMAU 400 mg group (P = .02), with a median increase of 22% (Fig. 2G, see Table 2). The change in serum BAP did not differ across treatment groups (P = .31).
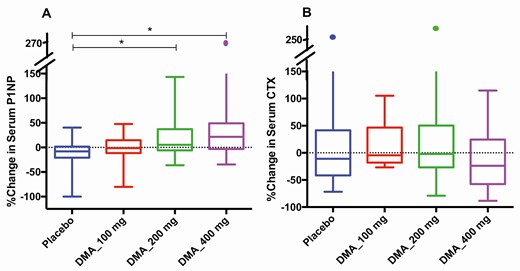
Comparison of percentage changes in bone turnover markers in various groups by end of treatment (median, interquartile range). This figure shows a box and whiskers plot of the percentage change in serum concentrations of A, procollagen type I amino-terminal propeptide (P1NP) and B, C-terminal telopeptide of type I collagen (CTX) by the end of treatment in various groups: placebo (blue), dimethandrolone (DMA) undecanoate (DMAU) 100 mg (red), DMAU 200 mg (green), and DMAU 400 mg (purple). *P less than .01 (significant pairwise comparisons) on Mann-Whitney U test, adjusted for multiple comparisons.
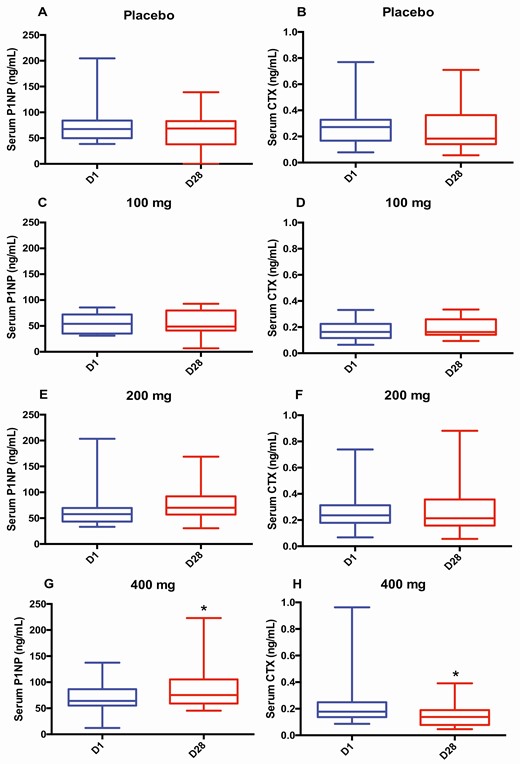
Serum bone turnover markers before and after treatment by group (median, interquartile range). This figure shows serum concentrations of procollagen type I amino-terminal propeptide (P1NP; A, C, E, and G) and C-terminal telopeptide of type I collagen (CTX; B, D, F, and H) in various groups: A and B, placebo; C and D, dimethandrolone undecanoate (DMAU) 100 mg; E and F, DMAU 200 mg; and G and H, DMAU 400 mg on day 1 (in blue; before treatment start) and day 28 (in red; end of treatment). *P less than .05 on Wilcoxon signed-rank test for within-group change.
Overall, change in serum CTX did not differ between any treatment groups (Fig. 1B) (P = .09). Within individual groups, serum CTX significantly declined from day 1 to day 28 only in the DMAU 400 mg group (P = .04), with a median decline of 24% (Fig. 2H, see Table 2).
Correlation of the bone turnover marker changes with hormones, androgenic end points, and drug concentrations
Change in P1NP was negatively correlated with change in serum T and serum LH on day 28 but not with change in serum E2 (Table 3). Similarly, change in P1NP negatively correlated with decreases in HDL-C and SHBG, but not with changes in serum Hct (see Table 3). Change in P1NP did not correlate with serum drug concentrations but there was a positive correlation with DMAU dose (see Table 3). Using stepwise reverse multivariate linear regression, only increase in weight and baseline P1NP were significantly associated with the increase in P1NP (P < .001, adjusted R2 = 0.34) when controlling for age, baseline P1NP, changes in serum T and E2, and dose of DMAU.
Correlation of change in serum procollagen type I amino-terminal propeptide with different variables
| Variable . | Spearman r . | P . |
|---|---|---|
| Change in serum T | –0.44 | < .001a |
| Change in serum E2 | –0.15 | .19 |
| Day 28 serum LHb | –0.43 | < .001a |
| Change in weight | 0.33 | .002a |
| Change in Hct | 0.13 | .26 |
| Change in HDL-C | –0.36 | .001a |
| Change in SHBG | –0.36 | .001a |
| Mean trough DMA | –0.16 | .16 |
| Day 28 CMax DMA | –0.02 | .87 |
| Day 28 AUC DMA | –0.02 | .88 |
| Dose of DMAU | 0.40 | < .001a |
| Variable . | Spearman r . | P . |
|---|---|---|
| Change in serum T | –0.44 | < .001a |
| Change in serum E2 | –0.15 | .19 |
| Day 28 serum LHb | –0.43 | < .001a |
| Change in weight | 0.33 | .002a |
| Change in Hct | 0.13 | .26 |
| Change in HDL-C | –0.36 | .001a |
| Change in SHBG | –0.36 | .001a |
| Mean trough DMA | –0.16 | .16 |
| Day 28 CMax DMA | –0.02 | .87 |
| Day 28 AUC DMA | –0.02 | .88 |
| Dose of DMAU | 0.40 | < .001a |
Change calculated as difference between first measurement on day 28 and day 1 of treatment.
Abbreviations:; AUC, area under the curve; CMax, maximum serum concentrations; DMA, dimethandrolone; DMAU, dimethandrolone undecanoate; E2, estradiol; Hct, hematocrit; HDL-C, high-density lipoprotein cholesterol; LH, luteinizing hormone; P1NP, procollagen type I amino-terminal propeptide; SHBG, sex-hormone binding globulin; T, testosterone.
aP less than .05 was significant.
bUsed pre-dose LH value on day 28 visit.
Correlation of change in serum procollagen type I amino-terminal propeptide with different variables
| Variable . | Spearman r . | P . |
|---|---|---|
| Change in serum T | –0.44 | < .001a |
| Change in serum E2 | –0.15 | .19 |
| Day 28 serum LHb | –0.43 | < .001a |
| Change in weight | 0.33 | .002a |
| Change in Hct | 0.13 | .26 |
| Change in HDL-C | –0.36 | .001a |
| Change in SHBG | –0.36 | .001a |
| Mean trough DMA | –0.16 | .16 |
| Day 28 CMax DMA | –0.02 | .87 |
| Day 28 AUC DMA | –0.02 | .88 |
| Dose of DMAU | 0.40 | < .001a |
| Variable . | Spearman r . | P . |
|---|---|---|
| Change in serum T | –0.44 | < .001a |
| Change in serum E2 | –0.15 | .19 |
| Day 28 serum LHb | –0.43 | < .001a |
| Change in weight | 0.33 | .002a |
| Change in Hct | 0.13 | .26 |
| Change in HDL-C | –0.36 | .001a |
| Change in SHBG | –0.36 | .001a |
| Mean trough DMA | –0.16 | .16 |
| Day 28 CMax DMA | –0.02 | .87 |
| Day 28 AUC DMA | –0.02 | .88 |
| Dose of DMAU | 0.40 | < .001a |
Change calculated as difference between first measurement on day 28 and day 1 of treatment.
Abbreviations:; AUC, area under the curve; CMax, maximum serum concentrations; DMA, dimethandrolone; DMAU, dimethandrolone undecanoate; E2, estradiol; Hct, hematocrit; HDL-C, high-density lipoprotein cholesterol; LH, luteinizing hormone; P1NP, procollagen type I amino-terminal propeptide; SHBG, sex-hormone binding globulin; T, testosterone.
aP less than .05 was significant.
bUsed pre-dose LH value on day 28 visit.
Discussion
DMAU, a potential once-daily “male contraceptive pill” with both androgenic and progestational activity, markedly suppresses serum gonadotropins, T and E2 (11). Despite near-castrate levels of T and E2 over 28 days of treatment, we found that serum P1NP, a marker of bone formation, increased in a dose-responsive manner with DMAU treatment. Notably, the change in serum BAP did not differ between treatment groups, even though the change in P1NP did. However, a discrepancy between changes in P1NP and BAP have been reported previously with a 3-week human trial manipulating the T/E2 balance, wherein there was no change in BAP despite a decline in P1NP (35). Even with the use of anabolic agents like teriparatide for osteoporosis, an increase in serum P1NP is observed more rapidly and with greater magnitude than changes in BAP (16, 36, 37). P1NP has also been shown to be a more sensitive marker of bone formation and more predictive of changes in BMD than BAP (38, 39). Serum CTX, a bone resorption marker, did not show a consistent change. These results raise the possibility that DMAU treatment may skew bone homeostasis toward increased bone formation in the short term without a significant change in resorption. However, longer-term studies of DMAU, in which multiple markers of bone formation and resorption as well as BMD are quantified, will only be able to define the true effect of DMAU on bone metabolism.
E2 is the main determinant of bone resorption in men (35, 40). Lower serum E2 concentrations in men have been shown to be associated with lower BMD (41) and increased fracture risk (42), independent of their serum T concentrations. Because the majority of circulating E2 in men comes from aromatization of T (43), serum concentrations of T and E2 are directly related in healthy men (44). In an elegant study using a combination of exogenous T and an aromatase inhibitor over 20 weeks of treatment, Finkelstein et al (45) observed a decline in BMD in the absence of E2, regardless of serum T concentrations. They suggested a threshold concentration of serum T (< 200 ng/dL) and E2 (< 10 pg/mL), below which a decline in BMD is likely to occur (45). Similar studies (35, 40) point to a critical role for circulating E2 in preserving bone health in men in the setting of normal or low serum androgen concentrations.
Studying changes in BTMs with sex-steroid manipulation also suggest that E2 plays a key role in downregulating bone resorption in men, while androgens contribute to bone formation. When circulating E2 concentrations decreased to less than 10 pg/mL in the Finkelstein study (45), serum CTX increased, despite normal serum androgen levels. In contrast, even at very low serum androgen concentrations (45), serum P1NP concentrations were maintained. Studies using a nonaromatizable but more potent (than T) androgen, dihydrotestosterone (DHT), showed no changes in bone formation in the short term (3 months) (46), an increase in serum P1NP in the long term (2 years) (28), and no change in bone resorption markers, despite serum E2 levels well below 10 pg/mL throughout the treatment period. Notably, by the end of 2 years, hip BMD was preserved, but spine BMD declined with DHT treatment (28). This dichotomy likely reflects the differential effects of sex steroids on trabecular and cortical bone (47, 48) and suggests that aromatization of androgens is important for preservation of trabecular BMD. Consistent with these observations, we observed that the highly androgenic steroid, DMAU, increased P1NP. We were intrigued to note that CTX was not significantly affected with administration of DMAU, despite marked suppression of serum E2 to less than 10 pg/ml.
ARs and estrogen receptors (ERs) are expressed on osteoblasts, osteoclasts, and osteocytes and can regulate bone remodeling through various mechanisms (49). Estrogen binding to ER on osteoclasts accounts for their protective effect on trabecular bone (50); however, it has been shown that ER on osteoblast progenitor cells does not require the presence of estrogens to promote cortical bone accrual (51). AR is highly expressed on osteocytes (52), which are the most abundant bone cells. Sclerostin is an important regulator of bone mass and prevents bone formation by osteoblasts (53). Treatment with the nonaromatizable androgen DHT downregulates sclerostin expression in osteocytes, and androgen action is an independent predictor of sclerostin production (54). Therefore, it is possible that DMA, like DHT, downregulates sclerostin production and thereby increases bone formation (and P1NP). Measurement of sclerostin following DMAU administration may be considered in future studies, but because there are no published data on stability of sclerostin in blood samples that are stored long term at –20°C, we did not measure it for this analysis.
The change in serum P1NP correlated with other pharmacodynamic end points (LH suppression, decreases in serum HDL-C and SHBG), suggesting that the observed changes were likely a result of the biological action of DMA. Interestingly, we found no correlation between changes in P1NP and serum drug concentrations, despite a correlation with dose of DMAU.
Participants receiving DMAU also gained weight in a dose-responsive manner, and this can affect bone turnover. In our multivariate analyses, weight remained independently associated with increases in P1NP, when adjusting for other variables (including DMAU dose), suggesting that DMA likely mediates at least some of its impacts on bone formation via weight gain. Data from obese individuals who lose weight suggest an early increase in CTX followed by long-term reduction in hip BMD (55, 56). In patients with anorexia nervosa, acute weight gain increases bone formation, reduces bone resorption, and increases BMD (57). However, studies are lacking demonstrating the impact of modest weight gain on BMD in healthy, eugonadal individuals. Increase in muscle mass and reduction in fat mass from androgen therapy could affect mechanical loading and bone metabolism.
The precise mechanism whereby DMAU increases P1NP in the short term is not clear. Although this may be related to its potent androgenicity, we cannot rule out an androgen-independent effect of higher doses of DMA on bone. The statistically significant decrease in serum CTX noted only with the DMAU 400-mg group also raises this possibility. Further in vitro studies may be warranted to understand the impact of DMAU on osteoclasts and osteoblasts.
Progesterone treatment, by binding to progesterone receptors (58), stimulates osteoblasts to create more bone matrix (59) and stimulates mesenchymal cells to form osteoblasts (60). While progestins may stimulate bone formation in women as long as bone resorption is maintained (61), their role in men’s bone health is unclear. Whether DMA binding to progesterone receptors may have contributed to the observed increases in P1NP warrants further investigation with in vitro and in vivo studies.
Despite being a retrospective secondary analysis, this study’s strengths include adequate power to interpret changes in BTMs, presence of a placebo group to compare changes, and accurate serum hormone measurements with highly sensitive liquid chromatography–mass spectrometry assays. However, our results need to be interpreted with caution because we are limited by the very short duration of drug treatment and we analyzed only one marker of bone resorption, and because the retrospective nature of this analysis prevented appropriate sample collection and accurate estimation of other well-defined bone markers (such as osteocalcin or TRAP5b [tartrate-resistant acid phosphatase 5b]) to corroborate our observations (62, 63)
We also cannot exclude the possibility of intraindividual variability in the value of BTMs because we ran the assay on only one sample at each time point. Future studies, with longer duration of treatment, will need to overcome these limitations and also include quantification of body composition and BMD to determine true effects of DMAU administration in healthy men.
Conclusion
In summary, we found that 28 days of treatment with a highly potent, nonaromatizable androgen, DMAU, in healthy men, resulted in increased serum P1NP, a bone formation marker, without significantly altering serum CTX, a marker of bone resorption. These results are intriguing, given existing literature suggesting that a threshold concentration of serum E2 may be required to prevent an increase in bone resorption in men. It is possible that at least in the short term, the high-potency androgenic activity of DMA alone or in combination with observed weight gain, overcomes E2 deficiency in preventing bone resorption. However, only longer studies can determine whether changes persist over time and the eventual impact of DMAU on bone health in men. Understanding the longer-term impact of DMAU on bone health will be critical in supporting the clinical development of DMAU as an oral male hormonal contraceptive.
Abbreviations
- AR
androgen receptor
- BAP
bone-specific alkaline phosphatase
- BMD
bone mineral density
- BTM
bone turnover markers
- CV
coefficient of variation
- DHT
dihydrotestosterone
- DMA
dimethandrolone
- DMAU
dimethandrolone undecanoate
- E2
estradiol
- ER
estrogen receptor
- Hct
hematocrit
- HDL-C
high-density lipoprotein cholesterol
- LH
luteinizing hormone
- P1NP
procollagen type I amino-terminal propeptide
- SHBG
sex-hormone binding globulin
- T
testosterone
Acknowledgments
We would like to thank all the study participants and Study Coordinators/Nurses Kathryn Torrez Duncan (University of Washington) and Xiaodan Han, and Elizabeth Ruiz (The Lundquist Institute, Harbor-UCLA) for their work conducting this study; Sarah Godfrey and Tricia Brady from Health Decisions for assistance with coordination and monitoring; Clint Dart from Health Decisions for input in data analysis; and Dr Min Lee, Contraceptive Development Program, Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), for supervision of manufacture and formulation of investigational drug product for the study.
Financial Support: This work is supported by the NICHD Contraceptive Clinical Trials Network under contracts with the University of Washington (Contract: HHSN275201300025I; Task Order: HHSN27500003), The Lundquist Institute at Harbor-UCLA Medical Center (Contract: HHSN27520130024I; Task Order: HHSN27500004), and Health Decisions (HHSN2752012002). F.Y. is supported by Eunice Kennedy Shriver National Institute of Child Health and Human Development (F32 HD097932). P.Y.L. is also supported in part by National Heart, Lung, and Blood Institute (K24HL13632). S.P. is also supported by the Robert McMillen Professorship in Lipid Research.
Clinical Trial Information: Clinical trial registration number: NCT01382069 (registered June 27, 2011).
Additional Information
Disclosure Summary: A.T., F.Y., A.N., and P.L.: have nothing to disclose. J.A. has served as a consultant for Clarus; R.S. has received grant support from Testosterone Replacement Therapy Manufacturer Consortium and has served as a consultant for Clarus; D.B. has served as principal investigator on a Cooperative Research and Development Agreement with HRA Pharma; C.W. has received grant support from TesoRx; and S.P. has served as a consultant for Clarus.
Data Availability
The data sets generated during and/or analyzed during the present study are not publicly available but are available from the corresponding author on reasonable request.
References



