-
PDF
- Split View
-
Views
-
Cite
Cite
Michaela Wilhelmi, Ulrich Franke, Tina Cohnert, Petra Weber, Jörg Kaukemüller, Stefan Fischer, Thorsten Wahlers, Axel Haverich, Coronary artery bypass grafting surgery without the routine application of blood products: is it feasible?, European Journal of Cardio-Thoracic Surgery, Volume 19, Issue 5, May 2001, Pages 657–661, https://doi.org/10.1016/S1010-7940(01)00648-0
Close - Share Icon Share
Abstract
Objective: Fresh frozen plasma (FFP) substitution is currently standard practise in cardiac surgery. In this study we investigate whether elective coronary artery bypass grafting (CABG) surgery is feasible without the administration of blood products compared to the substitution of fresh frozen plasma. Patients and methods: From September 1997 to April 1998, 120 consecutive patients underwent CABG surgery at our institution. In the FFP group patients (n=60; men, n=43; women, n=17; mean age: 64±8.5 years) received 4 units of fresh frozen plasma (FFP) intraoperatively. In comparison, patients in the control group (n=60; men, n=44; women, n=16; mean age: 65±7.5 years) did not receive FFP. Main endpoints included perioperative hematologic parameters, blood loss, and the amount of blood products that were administered. Results: Intraoperatively, the substitution of packed red blood cells (pRBC) in the FFP group was significantly higher compared with the control group (0.63±0.94 units/patient vs. 0.12±0.38 units/patient, P=0.001). Postoperatively, patients in the FFP group required significantly more pRBC products than patients in the control group (0.78±1.09 vs. 0.42±0.77, P=0.024). There were no significant differences in hemoglobin (FFP group: 99±11.1 g/dl; control group: 105±13.5 g/dl) and hematocrit levels (FFP group: 30±3.39%; control group: 32±4%). Also, no significant differences regarding the postoperative blood loss was observed between groups (FFP group: 588±224 vs. control group: 576±272 ml/24 h). Conclusions: This study clearly demonstrates that the avoidance of routine intraoperative FFP administration does not lead to an increase in blood loss postoperatively compared to patients that received FFPs. Furthermore, we did not observe increased requirements for postoperative FFP infusion in patients that did not receive FFPs intraoperatively.
1 Introduction
Coronary artery bypass grafting (CABG) is the most frequent procedure performed in cardiac surgery today. Apart from selected cases, the operation is usually performed under extracorporeal circulatory support. However, cardiopulmonary bypass (CPB) necessitates complete heparinization, which leads to increased blood loss and consequently to the administration of blood products. This is mainly due to impairment of the coagulation system by pump priming, heparinization, and the interaction between the blood and the artificial surface of the extracorporeal circuit.
Over the last decade attempts have been made to decrease the use of either full blood transfusion or packed red blood cells [19]. Instead, blood was collected and reintroduced into the patient by red cell saver systems [11]. However, with respect to the costs of blood preparation and transfusion as well as the risks of blood transfusion-related transmission of infectious diseases such as hepatitis or HIV, the transfusion of blood and blood products during CABG surgery needs to be evaluated.
The aim of this study was to examine whether routine CABG can be performed safely without any transfusion of blood or blood products as compared to the administration of fresh frozen plasma.
2 Patients and methods
Between September 1997 and April 1998, data from 120 consecutive patients undergoing primary coronary artery revascularization were recorded prospectively. Patients were block-randomized into groups of n=60 patients. All patients in the FFP group were operated on first, followed by the 60 control patients. This is an accurate approach since all procedures in this study were performed by two experienced cardiac surgeons. In the FFP group 60 patients received 4 units (250 cc each) of fresh frozen plasma (FFP) after completion of cardiopulmonary bypass, whereas in the control group 60 patients received 1000 cc of a hydroxy-ethyl-starch solution for equivalent volume substitution (HAES, Firma Braun Melsungen, Melsungen, Germany).
In order to provide a homogeneous patient cohort, all patients with previous thoracic surgical interventions, oral medication with thrombocyte aggregation inhibitors 72 h prior to surgery,history of coagulation disorders and a left ventricular ejection fraction of less than 40% were excluded from the study. Two patients who underwent emergency surgery without presenting any exclusion criteria were included in the control group.
We analyzed the costs for all blood products administered to the 120 patients in this study. The costs per unit of blood products was obtained from the Department of Blood Transfusion of the Hannover Medical School. Following intraindividual comparison the total cost of blood products was determined for each group.
2.1 Surgical procedure
All operations were performed by two experienced cardiac surgeons. In all cases the left internal mammary artery (IMA) was anastomosed to the coronary artery system and additional vein grafts were anastomosed individually [3]. After median sternotomy the internal mammary artery was dissected. Simultaneously, the greater saphenous vein was harvested. A standard dose of aprotinine (1 000 000 units) was added to the priming solution of the CPB circuit. After priming the circuit with 1500 cc 0.9% saline and a 0.45% glucose solution, 1000 cc Ringer's solution and 40 ml sodium bicarbonate (Braun Melsungen), CPB was activated after intravenous application of 350 IU heparin per kg body weight. Moderate systemic hypothermia (31–33°C) was used for CPB. During CPB, anticoagulation was monitored by repeated measurements of the activated clotting time (ACT). An ACT of 500 s was aimed for. An initial dose of 1000 cc of St. Thomas-II cardioplegic solution was infused via the cross-clamped ascending aorta. After 30 min of cross-clamping or with reappearance of cardiac electric activity, a second dose of 300 ml cardioplegic solution was infused. All distal anastomoses were completed during aortic cross-clamping; all proximal anastomoses were performed under partial clamping of the ascending aorta. Anticoagulation was antagonized after termination of extracorporeal circulation by protamine infusion (300 IU/kg body weight). During surgery intrathoracic blood was collected for retransfusion by a cell-saver system (C.A.T.S. AT1, Fresenius, Mannheim, Germany).
Patients in both groups received 4 units of FFP postoperatively if the cumulative blood loss via chest tubes was more than 200 ml during the first hour, 350 ml during the first 2 h and 500 ml during the first 4 h. Packed red blood cell units (300 ml each) were substituted only if the hemoglobin level decreased below 8.0 g/dl.
2.2 Assessment
The following parameters were measured preoperatively and 3, 6, 12, 24, and 48 h postoperatively as well as prior to discharge from hospital (postoperative days 5–9): hemoglobin (Hb), hematocrit (Hc), platelet count (pc), prothrombin time (PT), and activated partial thromboplastin time (aPTT).
Cumulative blood loss was assessed at 1, 4, and 24 h postoperatively. In addition, the total amount of retransfused cell-saver-blood was analyzed. Any substitution of blood or blood products until discharge was documented.
Assessment of perioperative myocardial ischemic events included the documentation of the appearance of Q waves or ST segment elevation in two consecutive 12-channel electrocardiograms (ECG), or significant postoperative increases of cardiac Troponin-I (≧12.5 μg/l 24 h), or creatine kinase isoenzyme MB (CKMB≫10 units/l) serum levels.
2.3 Statistical analysis
Statistical analysis was performed by using SPSS (Statistical package for social sciences) for Windows, Version 7.5.2 (SPSS Inc., Chicago, IL). Data are presented as mean±standard deviation. Quantitative variables were analyzed by the Chi-square test. Comparison of values between the two groups regarding blood loss, hemoglobin, hematocrit levels, platelet count, and retransfused blood was performed by a t-test for independent samples. Values within the groups during the observation period were compared by paired t-test. Differences were considered significant with at a P-value of less than 0.05.
3 Results
Both groups were similar with regard to age, gender, incidence of preoperative myocardial infarction incidence, time of surgery, cardiopulmonary bypass time, time of aortic cross-clamping, number of distal anastomoses per patient, and volume of cardioplegic solution, as well as volume of retransfused cell-saver blood, as shown in Table 1 .
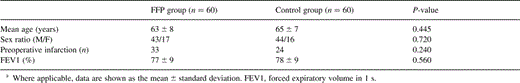
There were no deaths during the entire study period. No patient showed signs of a perioperative myocardial infarction. One patient in the FFP group (1.7%) and two patients in the control group (3.3%) underwent rethoracotomy for bleeding. In the FFP group, the mean cumulative blood loss during the first 6 h postoperatively was 400±155 and 588±224 ml after 24 h as compared to 348±194 and 576±272 ml in the control group, respectively. These differences were not significant. Cumulative blood loss for all patients within 24 h postoperatively ranged between 320 and 790 ml (median: FFP group 750 ml, control group 500 ml).
The postoperative stay in the intensive care unit (ICU) was equal in both groups (FFP group: 2±0.8 days vs. control group: 2±0.8 days). However, the postoperative ventilation time was significantly longer in the FFP group, 10±6.6 h, as compared to 8±3.8 h in the control group (P=0.03), as shown in Table 2 .
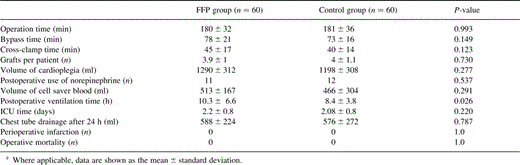
The partial thromboplastin time (aPTT) was similar in both groups comparing preoperative and 24-h postoperative values. At admission to the ICU patients in the FFP group showed a higher aPTT value of 59±7 s compared with 40±7 s in the control group. The platelet count did not differ significantly between both groups (Table 3) .
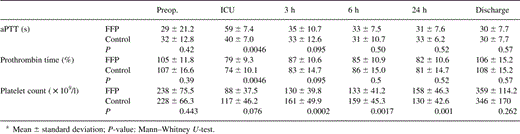
Also, no significant differences in postoperative hemoglobin blood levels comparing both groups were observed (Fig. 1) . However, hemoglobin and hematocrit blood levels decreased significantly in both groups comparing pre- and postoperative values (hemoglobin: FFP group: preop. 14.1±1.4 g/dl, postop. 10±0.8 g/dl; control group: preop. 13.9±1.5 g/dl, postop. 10.6±1.2 g/dl; hematocrit: FFP group: preop. 42.2±4.2%, postop. 33.5±6.3%; control group: preop. 41.3±4.3%, postop. 32.2±3.5%; P≪0.05 for each).
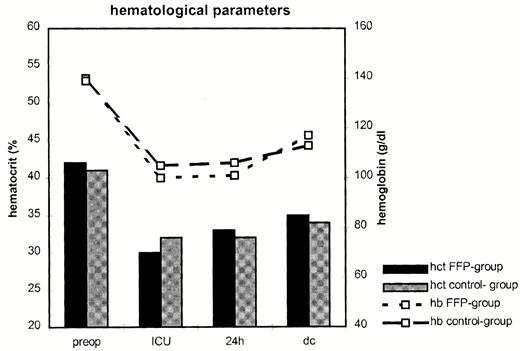
Hematological parameters during hospital stay in the FFP group and the control group. Preop, preoperative value; ICU, value at postoperative arrival at intensive care unit; dc, discharge; hct, hematocrit (%); hb, hemoglobin (g/dl).
Twenty-two patients (37%) in the FFP group required packed red blood cell units during their hospital stay compared with only seven patients (11%) in the control group (P=0.001) The intraoperative administration of packed red blood cell units was significantly higher in the FFP group compared with the control group (0.6±0.9 unit/patient vs. 0.1±0.4 unit/patient, P=0.001). The total amount of packed red blood cell units administered was 1.6±2 units/patient in the FFP cohort vs. 0.6±1 unit/patient in the control population (P=0.05).
The total amount of substituted blood plasma was 4±1 units in the FFP group and 0.2±0.7 unit in the control group (P=0.001).
4 Discussion
This study was performed to verify whether coronary artery bypass surgery is feasible without the routine application of fresh frozen plasma and without an increase in the risk for bleeding, substitution of packed red blood cells, and the overall complication rate.
The influence of cardiopulmonary bypass on hemostasis and coagulation is complex and well described in the literature [1]. In a study by Martinowitz and co-workers comparing the effect of fresh frozen plasma versus platelet substitution, the application of fresh frozen plasma did not influence bleeding time and platelet aggregation [1]. Previous studies demonstrated that the main cause of postoperative bleeding is platelet dysfunction after CPB in addition to the decrease of platelet counts and alterations in the coagulation cascade [4,5]. It is mainly the contact of blood with artificial surfaces that causes important alterations in hemostasis after CPB [6,7]. The intrinsic system is activated by contact activation through the artificial surface [7]. This results in a consumption of platelets and procoagulation factors by CPB [8]. Subsequently, CPB running periods of more than 2–3 h cause severe imbalances in the coagulation cascade [7,8]. Beside these mechanisms, hemodilution due to systemic application of the cardioplegic solution and the necessity of CPB circuit priming results in a further dilution as well as decrease in platelet function and reduction of hemostasis [3]. Platelet dysfunction as a result of mechanical shear forces in roller pump systems is an important cause of nonsurgical postoperative bleeding [3,9]. In addition, hypothermia has been shown to be able to induce platelet dysfunction [10].
To counteract platelet dysfunction investigators have used FFPs routinely during or following CPB with the intention to adjust coagulation factors, which are decreased due to hemodilution during CPB [1,4,5]. In our study only two patients in the FFP group required surgical reintervention for bleeding. Over the last years intra- and postoperative erythrocyte substitution has continually decreased while the use of FFP has increased [11].
The cumulative postoperative blood loss observed in our study did not differ between the two groups. The overall blood loss was considerably lower in our patient cohort compared with data reported by other groups [1–3]. Our results therefore demonstrate that a strategy without the routine substitution of fresh frozen plasma does not increase the risk of perioperative bleeding. Studies of Harker and co-workers [12] and Mammen and associates [13] support our results, regarding the lack of differences between the groups. Similar postoperative results were reported by Boldt and colleagues [10] and Consten and co-workers [3].
An additional aspect, which supports our current approach, is the potential risk of viral infection transmission by the transfusion of blood and blood products. Reports in the literature indicate an infection risk for hepatitis B of approximately 1/220 000 blood units, of 1/6210 to 1/40 000 blood units for hepatitis C, and of 11.6/100 000 to 1/800 000 blood units for HIV (by blood transfusion [14–16]). In addition, blood transfusion can induce severe and sometimes life-threatening allergic reactions [17].
Our observation, that patients in the FFP group more often seem to require additional FFP (11.4%) and pRBC (37%) substitution than patients in the control group (FFP 8.3%, RBC 12%) is surprising. Further mechanistic studies are needed to examine the underlying mechanisms of this phenomenon. However, this was not the aim of this study.
Analyzing the costs of treatment, there was an almost fourfold advantage in patients with infusion of colloid fluids. The costs for 500 cc HAES was 10 Euro compared with 41 Euro for one unit of FFP. The overall cost for blood products of all 60 patients in the FFP group was 15.142 Euro, which was seven times higher than in the FFP group with 2182 Euro. The cost per patient was 252 Euro in the FFP group and 36 Euro in the control group. Because of differences in individual hospital calculations and current exchange rates, a direct comparison with data reported in the literature is difficult [18].
In summary, we demonstrate that the avoidance of routine intraoperative administration of fresh frozen plasma is not associated with an increase in bleeding disorders or perioperative transfusion requirements in patients following routine CABG surgery. Furthermore, the potential risk of transmission of viral diseases is lower compared with conventional strategies such as the application of fresh frozen plasma. We therefore suggest that coronary artery bypass surgery is safe and cost-effective without the intraoperative transfusion of fresh frozen plasma. This approach should be considered for routine use in patients undergoing CABG without previous thoracic surgical interventions or coagulation disorders.




