-
PDF
- Split View
-
Views
-
Cite
Cite
Angelique E Boutzoukas, Angelique E Boutzoukas, Ryan Kilpatrick, Daniel Benjamin, Jennifer Le, Jennifer Le, Rachel Greenberg, Kelly Wade, Michael Cohen-Wolkowiez, Kanecia O Zimmerman, 565. Pilot Study of Prolonged Post-Discontinuation Antibiotic Exposure (PDAE) to Ampicillin Among Low Birthweight Preterm Infants, Open Forum Infectious Diseases, Volume 9, Issue Supplement_2, December 2022, ofac492.618, https://doi.org/10.1093/ofid/ofac492.618
Close - Share Icon Share
Abstract
In the United States, 75% of preterm infants receive ampicillin after birth for evaluation of early onset sepsis. Excess antibiotic exposure in preterm infants is associated with morbidity and mortality. Pharmacokinetic (PK) simulations suggest that very low birthweight (< 1500g) infants receiving ampicillin may experience excess and prolonged therapeutic exposures to ampicillin after drug discontinuation, called post-discontinuation antibiotic exposures (PDAE). It is unknown if low birthweight ( > 1500g to < 2500g) infants that receive ampicillin experience prolonged PDAE.
We conducted a pilot prospective PK clinical trial at a single tertiary care center. We enrolled infants with gestational age (GA) < 36 weeks and birthweight (BW) > 1500g and < 2500g who received ampicillin 200 mg/kg/day per standard of care. All infants received 6 ampicillin 66mg/kg doses administered Q8H within 24 hours following birth. Post-discontinuation PK samples were collected between 8-120 hours after the last dose of ampicillin. We performed descriptive statistics on patient characteristics, and graphically displayed ampicillin plasma concentrations following discontinuation. We defined therapeutic exposures as concentrations above the minimum inhibitory concentration (MIC) for Group B Streptococcus (0.25mcg/mL).
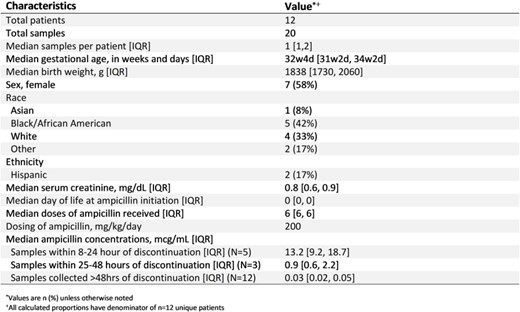
Enrolled Patient Characteristics and Plasma Ampicillin Concentrations
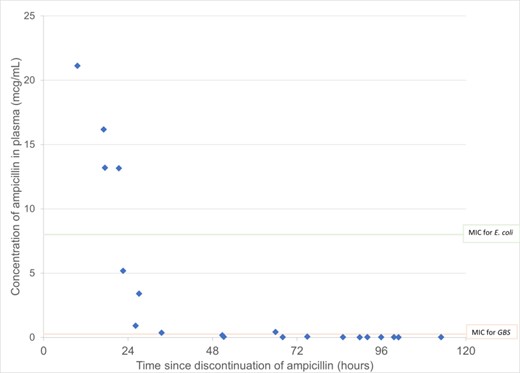
Ampicillin Post-Discontinuation Antibiotic Exposures in Low Birthweight Infants (1500g-2500g)
In this small cohort of premature infants, exposures of ampicillin remained therapeutic for at least 24 hours post drug discontinuation and suggests that shorter duration of empirical ampicillin treatments may be warranted in this population.
Daniel Benjamin, Jr., MD PhD MPH, Allergan: Advisor/Consultant|Melinta Therapuetics: Advisor/Consultant|Syneos Health: Advisor/Consultant Rachel Greenberg, MD, MB, MHS, Provepharm Inc: Advisor/Consultant|Tellus Therapeutics: Advisor/Consultant.
Author notes
Session: 58. Pediatric Antimicrobial Stewardship
Thursday, October 20, 2022: 12:15 PM
- ampicillin
- antibiotics
- plasma drug concentration
- exposure
- birth weight
- consultants
- disclosure
- gestational age
- infant
- low birth weight infant
- infant, premature
- infant, very low birth weight
- plasma
- morbidity
- mortality
- streptococcus group b
- antimicrobial susceptibility test
- minimum inhibitory concentration result
- minimum inhibitory concentration measurement
- tertiary care hospitals
- descriptive statistics
- standard of care
- symbols
- malnutrition-inflammation-cachexia syndrome
- early onset neonatal sepsis





Comments