-
PDF
- Split View
-
Views
-
Cite
Cite
Jennifer C. Hunter, Yi Mu, Ghinwa K. Dumyati, Monica M. Farley, Lisa G. Winston, Helen L. Johnston, James I. Meek, Rebecca Perlmutter, Stacy M. Holzbauer, Zintars G. Beldavs, Erin C. Phipps, John R. Dunn, Jessica A. Cohen, Johannetsy Avillan, Nimalie D. Stone, Dale N. Gerding, L. Clifford McDonald, Fernanda C. Lessa, Burden of Nursing Home-Onset Clostridium difficile Infection in the United States: Estimates of Incidence and Patient Outcomes, Open Forum Infectious Diseases, Volume 3, Issue 1, Winter 2016, ofv196, https://doi.org/10.1093/ofid/ofv196
Close - Share Icon Share
Abstract
Background. Approximately 4 million Americans receive nursing home (NH) care annually. Nursing home residents commonly have risk factors for Clostridium difficile infection (CDI), including advanced age and antibiotic exposures. We estimated national incidence of NH-onset (NHO) CDI and patient outcomes.
Methods. We identified NHO-CDI cases from population-based surveillance of 10 geographic areas in the United States. Cases were defined by C difficile-positive stool collected in an NH (or from NH residents in outpatient settings or ≤3 days after hospital admission) without a positive stool in the prior 8 weeks. Medical records were reviewed on a sample of cases. Incidence was estimated using regression models accounting for age and laboratory testing method; sampling weights were applied to estimate hospitalizations, recurrences, and deaths.
Results. A total of 3503 NHO-CDI cases were identified. Among 262 sampled cases, median age was 82 years, 76% received antibiotics in the 12 weeks prior to the C difficile-positive specimen, and 57% were discharged from a hospital in the month before specimen collection. After adjusting for age and testing method, the 2012 national estimate for NHO-CDI incidence was 112 800 cases (95% confidence interval [CI], 93 400–131 800); 31 400 (28%) were hospitalized within 7 days after a positive specimen (95% CI, 25 500–37 300), 20 900 (19%) recurred within 14–60 days (95% CI, 14 600–27 100), and 8700 (8%) died within 30 days (95% CI, 6600–10 700).
Conclusions. Nursing home onset CDI is associated with substantial morbidity and mortality. Strategies focused on infection prevention in NHs and appropriate antibiotic use in both NHs and acute care settings may decrease the burden of NHO CDI.
Clostridium difficile is the most common cause of healthcare-associated infectious diarrhea and contributes to substantial morbidity, mortality, and medical costs among hospitalized patients [1]. Clostridium difficile infections (CDIs) are not restricted to hospital settings and have been increasingly recognized in community and nursing home settings [2]. In the United States, C difficile was responsible for close to a half a million infections in 2011 and is considered one of the nation's most urgent antimicrobial resistant threats [3, 4]. Approximately one quarter of CDI cases have symptom onset in nursing homes [4]; these residents have multiple risk factors associated with CDI including their often advanced age, increased healthcare utilization, extended length of stay, underlying conditions, and antimicrobial exposure [5]. Clostridium difficile infection incidence in persons 65 years of age and older is at least 5 times that of younger people, and advanced age is an important risk factor for recurrent CDI and related to changes in intestinal microbiota associated with greater risk of complicated CDI [4–8]. Of the estimated 29 000 deaths within 30 days of CDI diagnosis in 2011, approximately 90% were in those 65 years of age and older [4].
Nursing homes are an important healthcare setting for examining the CDI epidemiology and disease burden among the older population. Approximately 4 million people in the United States receive care every year in more than 15 000 nursing homes [9]. Residents of nursing homes are at increased risk for CDI from healthcare exposures that occur during and prior to staying in the nursing home, particularly because residents commonly move across healthcare settings [10]; according to data from the 2004 National Nursing Home Survey, approximately 40% of nursing home residents were in an acute care hospital before admission [11], and a previous study found that approximately half of nursing home-onset (NHO) CDI occurred in patients with recent hospitalization [12]. Although incidence of NHO CDI has been assessed [4, 12–14], national estimates of NHO-CDI hospitalization, death, and recurrence are not well described. Understanding the burden of CDI in nursing homes is critical for evaluating the effectiveness of prevention measures, including implementation of antimicrobial stewardship and infection control programs, and for identifying changes in epidemiology and clinical outcomes of CDI [15]. We used 2012 CDI population-based surveillance data from 10 US geographic areas to estimate national incidence, recurrence, hospitalization, and death among patients with onset of CDI in nursing homes and describe the demographic and clinical characteristics of these individuals.
METHODS
Clostridium difficile Infection Surveillance
Clostridium difficile infection surveillance is a component of the Centers for Disease Control and Prevention (CDC)'s Emerging Infections Program (EIP). In 2012, active population- and laboratory-based CDI surveillance was conducted in 35 counties in 10 states (California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee), encompassing 348 nursing homes and approximately 11.4 million people. The surveillance methods have been described previously [16]. In brief, surveillance personnel at participating sites investigate reports of all stool specimens positive for C difficile by either toxin or molecular assays from inpatient and outpatient laboratories serving surveillance area residents. A CDI case was defined as a surveillance area resident at least 1 year of age from whom a C difficile-positive stool was collected from January 1, 2012 through December 31, 2012 without a positive test in the prior eight weeks (ie, incident case) [16]. Cases were defined as NHO CDI if the C difficile-positive specimen was collected in a nursing home or from a nursing home resident either in an outpatient setting or within 3 days of hospital admission.
The EIP CDI surveillance has a sampling scheme that has been described previously [17]. In brief, 8 of the 10 EIP surveillance sites conduct an initial medical record review for all CDI cases, including basic demographic information, location, and date of C difficile-positive stool collection, hospitalization within 7 days after stool collection, and place of residence prior to stool collection. In the remaining 2 EIP sites (Georgia and Colorado), which have the largest surveillance populations, an initial medical record review is performed on a stratified random sample of CDI cases [4]. Based on the initial record review, a full medical record review is performed for 1 of every 10 healthcare-facility onset cases [4]. The full medical review includes collection of additional data on demographics, clinical findings, healthcare, and medication exposure in the 12 weeks prior to C difficile-positive stool collection, and outcomes, including recurrence and death.
Outcomes of Interest and Definitions
We defined 4 outcomes of interest: NHO-CDI incidence, hospitalization within 7 days, recurrence within 14–60 days, and death within 30 days after positive specimen collection. Incidence was calculated as the number of persons meeting the NHO-CDI case definition in the surveillance site divided by the total population for that surveillance site in 2012. Only the first recurrent CDI episode for each case was captured.
Nursing homes include freestanding physical rehabilitation, skilled nursing, and inpatient hospice facilities; nursing home residence and location of stool collection was determined through laboratory and medical record review.
Additional Data Sources
The 2012 Health Resources and Services Administration Area Health Resource Files were used to obtain county-level information on healthcare utilization, including the average number of inpatient-days per hospital, percentage of population living in nursing homes, and the total number of skilled nursing facility beds [18]. Population estimates were obtained from the 2012 US Census.
Data on laboratory testing methods for C difficile diagnosis were obtained through a survey of all the 139 inpatient and outpatient laboratories serving the EIP surveillance areas. Nucleic acid amplification test (NAAT) usage was defined as the proportion of 2012 CDI cases identified by NAAT as either the first- or second-line test.
Statistical Analysis
Data were analyzed using SAS software (version 9.3; SAS Institute). CDI cases with missing race (23%), including sampled cases from Georgia and Colorado, had variables imputed based on the known distribution of race by age, sex, and surveillance site. After race imputation was performed, a domain (subpopulation) analysis was used to estimate the number of CDI cases that were NHO in EIP sites where sampling was performed (Georgia and Colorado).
To generate the NHO-CDI national incidence estimate, a generalized linear mixed model with negative binomial distribution was built. The initial model included predictors previously demonstrated to be associated with CDI incidence, including age group (1–64, 65–84, ≥85 years), sex, race, NAAT usage, average inpatient-days per hospital, percentage of population in nursing homes, and total number of skilled nursing facility beds in each surveillance site. Predictors significantly associated with NHO-CDI incidence across EIP sites were retained in the final model, using a stay criterion of P ≤ .05. Estimated national NHO-CDI case counts were calculated based on model coefficients accounting for age of the US population and NAAT usage (ie, proportion of CDI cases identified by NAAT) across EIP sites because US NAAT usage is unknown. The 95% confidence intervals (CIs) for the NHO-CDI national estimates by each epidemiologic category were constructed based on imputation error, modeling error, and sampling error for Georgia and Colorado, where sampling was performed.
National estimates and corresponding 95% CIs for hospitalization, recurrence, and death within 30 days were calculated using 2 steps: (1) domain analysis to account for sampling design across EIP sites and (2) EIP site and national sampling weights for the national projections.
Site-specific and national population-based CDI incidence were calculated using 2012 US Census data. Infants <1 year of age were excluded from the denominator because they were not included in the numerator.
Laboratory Testing
A convenience sample of laboratories sent stool specimens from incident NHO-CDI cases with a full medical record review to reference laboratories for C difficile isolation [19]. Recovered isolates were sent to the CDC for molecular characterization using capillary gel electrophoresis-based polymerase chain reaction ribotyping [20].
Human Subjects
The EIP CDI surveillance was approved by the institutional review boards at the CDC and participating surveillance sites.
RESULTS
Case Characteristics
A total of 16 449 CDI cases were identified in 2012. Of these, 3503 (21%), representing 3320 patients, were estimated to be NHO CDI; 158 patients (5%) had more than 1 NHO-CDI case, including 136 with 2 incident episodes, 19 patients with 3 incident episodes, and 3 patients with 4 incident episodes. Of the 2567 NHO-CDI cases that had at least an initial medical record review performed, the median age was 81 years (range, 1–106 years), 85% were 65 years or older, and only 3 were <17 years. The majority of cases were female (60%) or white (63%). Sixty-nine percent of NHO-CDI cases had the positive C difficile stool specimen collected in a nursing home, whereas 31% were nursing home residents who had stool collected in an outpatient setting (eg, emergency department) or within the first 3 days of hospital admission.
Among 262 cases with full medical record review, 76% had diarrhea documented in the medical record (Table 1). Nursing homes-onset CDI cases frequently had underlying conditions, most commonly diabetes, dementia, and congestive heart failure. More than three quarters of NHO-CDI cases had documented receipt of antibiotics in the 12 weeks prior to the C difficile-positive specimen, primarily fluoroquinolones, β-lactam/β-lactamase inhibitor combinations, glycopeptides (vancomycin), or 3rd- or 4th-generation cephalosporins (Table 2). Of those cases with no documented prior antimicrobial therapy, 35% had exposure to a proton pump inhibitor or H-2 blocker in the 12 weeks prior to the C difficile-positive specimen collection.
Clinical Characteristics of NHO-CDI Cases With Full Medical Record Review (n = 262)
| Variable . | Residents (n = 262) . | |
|---|---|---|
| n . | % . | |
| Clinical findings | ||
| Diarrhea (per medical record documentation) | 199 | 76.0 |
| White blood cell count >15 000/µL | 53 | 20.2 |
| Underlying condition | ||
| Any underlying condition | 233 | 88.9 |
| Diabetes | 101 | 38.5 |
| Dementia | 76 | 29.0 |
| Congestive Heart Failure | 63 | 24.0 |
| CVA/Stroke | 55 | 21.0 |
| Chronic Pulmonary Disease | 58 | 22.1 |
| Chronic Renal Insufficiency | 51 | 19.5 |
| Peripheral Vascular Disease | 26 | 9.9 |
| Solid tumor (nonmetastatic) | 21 | 8.0 |
| Myocardial Infarct | 21 | 8.0 |
| Diverticular Disease | 18 | 6.9 |
| Hemiplegia/Paraplegia | 13 | 5.0 |
| Metastatic Solid Tumor | 10 | 3.8 |
| Connective Tissue Disease | 8 | 3.1 |
| Chronic Liver Disease | 7 | 2.7 |
| Peptic Ulcer Disease | 6 | 2.3 |
| Hematologic Malignancy | 5 | 1.9 |
| Inflammatory Bowel Disease | 2 | 0.8 |
| HIV | 1 | 0.4 |
| AIDS or CD4 count <200 | 1 | 0.4 |
| Unknown | 5 | 1.9 |
| None | 24 | 9.2 |
| Healthcare exposures in the 12 wks prior to incident Clostridium difficile-positive stool collection | ||
| Hospitalized overnight | 200 | 76.3 |
| Surgical procedure (inpatient or ambulatory surgery center) | 69 | 26.3 |
| Emergency room visit | 60 | 22.9 |
| Chronic hemodialysis | 11 | 4.2 |
| Observation/clinical decision unit stay | 5 | 1.9 |
| Medications in the 12 wks prior to incident C difficile-positive stool collection | ||
| Immunosuppressive Therapy | ||
| Any immunosuppressive therapy | 40 | 15.3 |
| Steroids | 32 | 12.2 |
| Chemotherapy | 8 | 3.1 |
| Other agents | 3 | 1.1 |
| No immunosuppressive therapy | 206 | 78.6 |
| Unknown immunosuppressive therapy | 16 | 6.1 |
| Proton Pump Inhibitor or H2-Blocker | ||
| Proton pump inhibitor | 120 | 45.8 |
| H2 blocker | 33 | 12.6 |
| Antimicrobial Therapy | ||
| Any antimicrobial therapy | 200 | 76.3 |
| No antimicrobial therapy | 46 | 17.6 |
| Antimicrobial therapy unknown | 16 | 6.1 |
| Antimicrobial Therapy, Proton Pump Inhibitor, or H2-Blocker | ||
| Any antimicrobial therapy, proton pump inhibitor, or H2-blocker | 228 | 87.0 |
| Exposure to a proton pump inhibitor or H2-blocker among patients with no antimicrobial therapy | 22 | 35 |
| Patient outcomes | ||
| Survived | 232 | 88.5 |
| Died | 25 | 9.5 |
| Recurrencea | 53 | 20.2 |
| Previous unique CDI episode (>8 wks prior to incident episode) | 41 | 15.6 |
| Patient admitted to hospital due to CDI (within 7 d of stool collection) | 32 | 12.2 |
| ICU admission | 6 | 2.3 |
| Toxic Megacolon or Ileus | 4 | 1.5 |
| Colectomy | 1 | 0.4 |
| Pseudomembranous colitis | 0 | 0.0 |
| Variable . | Residents (n = 262) . | |
|---|---|---|
| n . | % . | |
| Clinical findings | ||
| Diarrhea (per medical record documentation) | 199 | 76.0 |
| White blood cell count >15 000/µL | 53 | 20.2 |
| Underlying condition | ||
| Any underlying condition | 233 | 88.9 |
| Diabetes | 101 | 38.5 |
| Dementia | 76 | 29.0 |
| Congestive Heart Failure | 63 | 24.0 |
| CVA/Stroke | 55 | 21.0 |
| Chronic Pulmonary Disease | 58 | 22.1 |
| Chronic Renal Insufficiency | 51 | 19.5 |
| Peripheral Vascular Disease | 26 | 9.9 |
| Solid tumor (nonmetastatic) | 21 | 8.0 |
| Myocardial Infarct | 21 | 8.0 |
| Diverticular Disease | 18 | 6.9 |
| Hemiplegia/Paraplegia | 13 | 5.0 |
| Metastatic Solid Tumor | 10 | 3.8 |
| Connective Tissue Disease | 8 | 3.1 |
| Chronic Liver Disease | 7 | 2.7 |
| Peptic Ulcer Disease | 6 | 2.3 |
| Hematologic Malignancy | 5 | 1.9 |
| Inflammatory Bowel Disease | 2 | 0.8 |
| HIV | 1 | 0.4 |
| AIDS or CD4 count <200 | 1 | 0.4 |
| Unknown | 5 | 1.9 |
| None | 24 | 9.2 |
| Healthcare exposures in the 12 wks prior to incident Clostridium difficile-positive stool collection | ||
| Hospitalized overnight | 200 | 76.3 |
| Surgical procedure (inpatient or ambulatory surgery center) | 69 | 26.3 |
| Emergency room visit | 60 | 22.9 |
| Chronic hemodialysis | 11 | 4.2 |
| Observation/clinical decision unit stay | 5 | 1.9 |
| Medications in the 12 wks prior to incident C difficile-positive stool collection | ||
| Immunosuppressive Therapy | ||
| Any immunosuppressive therapy | 40 | 15.3 |
| Steroids | 32 | 12.2 |
| Chemotherapy | 8 | 3.1 |
| Other agents | 3 | 1.1 |
| No immunosuppressive therapy | 206 | 78.6 |
| Unknown immunosuppressive therapy | 16 | 6.1 |
| Proton Pump Inhibitor or H2-Blocker | ||
| Proton pump inhibitor | 120 | 45.8 |
| H2 blocker | 33 | 12.6 |
| Antimicrobial Therapy | ||
| Any antimicrobial therapy | 200 | 76.3 |
| No antimicrobial therapy | 46 | 17.6 |
| Antimicrobial therapy unknown | 16 | 6.1 |
| Antimicrobial Therapy, Proton Pump Inhibitor, or H2-Blocker | ||
| Any antimicrobial therapy, proton pump inhibitor, or H2-blocker | 228 | 87.0 |
| Exposure to a proton pump inhibitor or H2-blocker among patients with no antimicrobial therapy | 22 | 35 |
| Patient outcomes | ||
| Survived | 232 | 88.5 |
| Died | 25 | 9.5 |
| Recurrencea | 53 | 20.2 |
| Previous unique CDI episode (>8 wks prior to incident episode) | 41 | 15.6 |
| Patient admitted to hospital due to CDI (within 7 d of stool collection) | 32 | 12.2 |
| ICU admission | 6 | 2.3 |
| Toxic Megacolon or Ileus | 4 | 1.5 |
| Colectomy | 1 | 0.4 |
| Pseudomembranous colitis | 0 | 0.0 |
Abbreviations: AIDS, acquired immune deficiency syndrome; CDI, Clostridium difficile infection; CVA, cerebrovascular accident; HIV, human immunodeficiency virus; ICU, intensive care unit; NHO, nursing home onset.
a Recurrence was defined as a C difficile-positive stool specimen between 2 and 8 weeks after the last positive specimen collection. Only the first recurrent CDI episode for each case was captured.
Clinical Characteristics of NHO-CDI Cases With Full Medical Record Review (n = 262)
| Variable . | Residents (n = 262) . | |
|---|---|---|
| n . | % . | |
| Clinical findings | ||
| Diarrhea (per medical record documentation) | 199 | 76.0 |
| White blood cell count >15 000/µL | 53 | 20.2 |
| Underlying condition | ||
| Any underlying condition | 233 | 88.9 |
| Diabetes | 101 | 38.5 |
| Dementia | 76 | 29.0 |
| Congestive Heart Failure | 63 | 24.0 |
| CVA/Stroke | 55 | 21.0 |
| Chronic Pulmonary Disease | 58 | 22.1 |
| Chronic Renal Insufficiency | 51 | 19.5 |
| Peripheral Vascular Disease | 26 | 9.9 |
| Solid tumor (nonmetastatic) | 21 | 8.0 |
| Myocardial Infarct | 21 | 8.0 |
| Diverticular Disease | 18 | 6.9 |
| Hemiplegia/Paraplegia | 13 | 5.0 |
| Metastatic Solid Tumor | 10 | 3.8 |
| Connective Tissue Disease | 8 | 3.1 |
| Chronic Liver Disease | 7 | 2.7 |
| Peptic Ulcer Disease | 6 | 2.3 |
| Hematologic Malignancy | 5 | 1.9 |
| Inflammatory Bowel Disease | 2 | 0.8 |
| HIV | 1 | 0.4 |
| AIDS or CD4 count <200 | 1 | 0.4 |
| Unknown | 5 | 1.9 |
| None | 24 | 9.2 |
| Healthcare exposures in the 12 wks prior to incident Clostridium difficile-positive stool collection | ||
| Hospitalized overnight | 200 | 76.3 |
| Surgical procedure (inpatient or ambulatory surgery center) | 69 | 26.3 |
| Emergency room visit | 60 | 22.9 |
| Chronic hemodialysis | 11 | 4.2 |
| Observation/clinical decision unit stay | 5 | 1.9 |
| Medications in the 12 wks prior to incident C difficile-positive stool collection | ||
| Immunosuppressive Therapy | ||
| Any immunosuppressive therapy | 40 | 15.3 |
| Steroids | 32 | 12.2 |
| Chemotherapy | 8 | 3.1 |
| Other agents | 3 | 1.1 |
| No immunosuppressive therapy | 206 | 78.6 |
| Unknown immunosuppressive therapy | 16 | 6.1 |
| Proton Pump Inhibitor or H2-Blocker | ||
| Proton pump inhibitor | 120 | 45.8 |
| H2 blocker | 33 | 12.6 |
| Antimicrobial Therapy | ||
| Any antimicrobial therapy | 200 | 76.3 |
| No antimicrobial therapy | 46 | 17.6 |
| Antimicrobial therapy unknown | 16 | 6.1 |
| Antimicrobial Therapy, Proton Pump Inhibitor, or H2-Blocker | ||
| Any antimicrobial therapy, proton pump inhibitor, or H2-blocker | 228 | 87.0 |
| Exposure to a proton pump inhibitor or H2-blocker among patients with no antimicrobial therapy | 22 | 35 |
| Patient outcomes | ||
| Survived | 232 | 88.5 |
| Died | 25 | 9.5 |
| Recurrencea | 53 | 20.2 |
| Previous unique CDI episode (>8 wks prior to incident episode) | 41 | 15.6 |
| Patient admitted to hospital due to CDI (within 7 d of stool collection) | 32 | 12.2 |
| ICU admission | 6 | 2.3 |
| Toxic Megacolon or Ileus | 4 | 1.5 |
| Colectomy | 1 | 0.4 |
| Pseudomembranous colitis | 0 | 0.0 |
| Variable . | Residents (n = 262) . | |
|---|---|---|
| n . | % . | |
| Clinical findings | ||
| Diarrhea (per medical record documentation) | 199 | 76.0 |
| White blood cell count >15 000/µL | 53 | 20.2 |
| Underlying condition | ||
| Any underlying condition | 233 | 88.9 |
| Diabetes | 101 | 38.5 |
| Dementia | 76 | 29.0 |
| Congestive Heart Failure | 63 | 24.0 |
| CVA/Stroke | 55 | 21.0 |
| Chronic Pulmonary Disease | 58 | 22.1 |
| Chronic Renal Insufficiency | 51 | 19.5 |
| Peripheral Vascular Disease | 26 | 9.9 |
| Solid tumor (nonmetastatic) | 21 | 8.0 |
| Myocardial Infarct | 21 | 8.0 |
| Diverticular Disease | 18 | 6.9 |
| Hemiplegia/Paraplegia | 13 | 5.0 |
| Metastatic Solid Tumor | 10 | 3.8 |
| Connective Tissue Disease | 8 | 3.1 |
| Chronic Liver Disease | 7 | 2.7 |
| Peptic Ulcer Disease | 6 | 2.3 |
| Hematologic Malignancy | 5 | 1.9 |
| Inflammatory Bowel Disease | 2 | 0.8 |
| HIV | 1 | 0.4 |
| AIDS or CD4 count <200 | 1 | 0.4 |
| Unknown | 5 | 1.9 |
| None | 24 | 9.2 |
| Healthcare exposures in the 12 wks prior to incident Clostridium difficile-positive stool collection | ||
| Hospitalized overnight | 200 | 76.3 |
| Surgical procedure (inpatient or ambulatory surgery center) | 69 | 26.3 |
| Emergency room visit | 60 | 22.9 |
| Chronic hemodialysis | 11 | 4.2 |
| Observation/clinical decision unit stay | 5 | 1.9 |
| Medications in the 12 wks prior to incident C difficile-positive stool collection | ||
| Immunosuppressive Therapy | ||
| Any immunosuppressive therapy | 40 | 15.3 |
| Steroids | 32 | 12.2 |
| Chemotherapy | 8 | 3.1 |
| Other agents | 3 | 1.1 |
| No immunosuppressive therapy | 206 | 78.6 |
| Unknown immunosuppressive therapy | 16 | 6.1 |
| Proton Pump Inhibitor or H2-Blocker | ||
| Proton pump inhibitor | 120 | 45.8 |
| H2 blocker | 33 | 12.6 |
| Antimicrobial Therapy | ||
| Any antimicrobial therapy | 200 | 76.3 |
| No antimicrobial therapy | 46 | 17.6 |
| Antimicrobial therapy unknown | 16 | 6.1 |
| Antimicrobial Therapy, Proton Pump Inhibitor, or H2-Blocker | ||
| Any antimicrobial therapy, proton pump inhibitor, or H2-blocker | 228 | 87.0 |
| Exposure to a proton pump inhibitor or H2-blocker among patients with no antimicrobial therapy | 22 | 35 |
| Patient outcomes | ||
| Survived | 232 | 88.5 |
| Died | 25 | 9.5 |
| Recurrencea | 53 | 20.2 |
| Previous unique CDI episode (>8 wks prior to incident episode) | 41 | 15.6 |
| Patient admitted to hospital due to CDI (within 7 d of stool collection) | 32 | 12.2 |
| ICU admission | 6 | 2.3 |
| Toxic Megacolon or Ileus | 4 | 1.5 |
| Colectomy | 1 | 0.4 |
| Pseudomembranous colitis | 0 | 0.0 |
Abbreviations: AIDS, acquired immune deficiency syndrome; CDI, Clostridium difficile infection; CVA, cerebrovascular accident; HIV, human immunodeficiency virus; ICU, intensive care unit; NHO, nursing home onset.
a Recurrence was defined as a C difficile-positive stool specimen between 2 and 8 weeks after the last positive specimen collection. Only the first recurrent CDI episode for each case was captured.
NHO-CDI Cases With Antimicrobial Therapy in 12 Weeks Prior to Positive Clostridium difficile Stool Collection (n = 200)
| Antimicrobial Therapy (12 wks Before Stool Collection) . | n . | %a . |
|---|---|---|
| Any antimicrobial therapy | 200 | 76 |
| Fluoroquinolones | 78 | 30 |
| Ciprofloxacin | 51 | |
| Levofloxacin | 20 | |
| Moxifloxacin | 10 | |
| β-lactam/β-lactamase inhibitor combinations | 68 | 26 |
| Piperacillin-tazobactam | 45 | |
| Amoxicillin/clavulanic acid | 25 | |
| Ampicillin/sulbactam | 6 | |
| Glycopeptide | 55 | 21 |
| Vancomycin (IV) | 55 | |
| 3rd- and 4th-generation cephalosporins | 52 | 20 |
| Ceftriaxone | 39 | |
| Cefepime | 14 | |
| Cefpodoxime | 3 | |
| Ceftazidime | 1 | |
| Ceftizoxime | 1 | |
| 1st- and 2nd-generation cephalosporins | 33 | 13 |
| Cephalexin | 21 | |
| Cefazolin | 15 | |
| Cefotetan | 1 | |
| Macrolides | 23 | 9 |
| Azithromycin | 21 | |
| Erythromycin | 2 | |
| Clarithromycin | 1 | |
| Sulfa | 17 | 6 |
| Trimethoprim-sulfamethoxazole | 17 | |
| Carbapenems | 14 | 5 |
| Ertapenem | 7 | |
| Meropenem | 5 | |
| Imipenem | 2 | |
| Lincosamide | 13 | 5 |
| Clindamycin | 13 | |
| Penicillins | 9 | 3 |
| Amoxicillin | 7 | |
| Aminoglycosides | 4 | 1 |
| Gentamicin | 4 | |
| Otherb | 43 | 16 |
| Antimicrobial Therapy (12 wks Before Stool Collection) . | n . | %a . |
|---|---|---|
| Any antimicrobial therapy | 200 | 76 |
| Fluoroquinolones | 78 | 30 |
| Ciprofloxacin | 51 | |
| Levofloxacin | 20 | |
| Moxifloxacin | 10 | |
| β-lactam/β-lactamase inhibitor combinations | 68 | 26 |
| Piperacillin-tazobactam | 45 | |
| Amoxicillin/clavulanic acid | 25 | |
| Ampicillin/sulbactam | 6 | |
| Glycopeptide | 55 | 21 |
| Vancomycin (IV) | 55 | |
| 3rd- and 4th-generation cephalosporins | 52 | 20 |
| Ceftriaxone | 39 | |
| Cefepime | 14 | |
| Cefpodoxime | 3 | |
| Ceftazidime | 1 | |
| Ceftizoxime | 1 | |
| 1st- and 2nd-generation cephalosporins | 33 | 13 |
| Cephalexin | 21 | |
| Cefazolin | 15 | |
| Cefotetan | 1 | |
| Macrolides | 23 | 9 |
| Azithromycin | 21 | |
| Erythromycin | 2 | |
| Clarithromycin | 1 | |
| Sulfa | 17 | 6 |
| Trimethoprim-sulfamethoxazole | 17 | |
| Carbapenems | 14 | 5 |
| Ertapenem | 7 | |
| Meropenem | 5 | |
| Imipenem | 2 | |
| Lincosamide | 13 | 5 |
| Clindamycin | 13 | |
| Penicillins | 9 | 3 |
| Amoxicillin | 7 | |
| Aminoglycosides | 4 | 1 |
| Gentamicin | 4 | |
| Otherb | 43 | 16 |
Abbreviations: CDI, Clostridium difficile infection; IV, intravenous; NHO, nursing home onset.
a Percentage of all cases with a full medical record review, n = 262.
b Other antibiotics include the following: metronidazole, doxycycline, linezolid, nitrofurantoin, daptomycin.
NHO-CDI Cases With Antimicrobial Therapy in 12 Weeks Prior to Positive Clostridium difficile Stool Collection (n = 200)
| Antimicrobial Therapy (12 wks Before Stool Collection) . | n . | %a . |
|---|---|---|
| Any antimicrobial therapy | 200 | 76 |
| Fluoroquinolones | 78 | 30 |
| Ciprofloxacin | 51 | |
| Levofloxacin | 20 | |
| Moxifloxacin | 10 | |
| β-lactam/β-lactamase inhibitor combinations | 68 | 26 |
| Piperacillin-tazobactam | 45 | |
| Amoxicillin/clavulanic acid | 25 | |
| Ampicillin/sulbactam | 6 | |
| Glycopeptide | 55 | 21 |
| Vancomycin (IV) | 55 | |
| 3rd- and 4th-generation cephalosporins | 52 | 20 |
| Ceftriaxone | 39 | |
| Cefepime | 14 | |
| Cefpodoxime | 3 | |
| Ceftazidime | 1 | |
| Ceftizoxime | 1 | |
| 1st- and 2nd-generation cephalosporins | 33 | 13 |
| Cephalexin | 21 | |
| Cefazolin | 15 | |
| Cefotetan | 1 | |
| Macrolides | 23 | 9 |
| Azithromycin | 21 | |
| Erythromycin | 2 | |
| Clarithromycin | 1 | |
| Sulfa | 17 | 6 |
| Trimethoprim-sulfamethoxazole | 17 | |
| Carbapenems | 14 | 5 |
| Ertapenem | 7 | |
| Meropenem | 5 | |
| Imipenem | 2 | |
| Lincosamide | 13 | 5 |
| Clindamycin | 13 | |
| Penicillins | 9 | 3 |
| Amoxicillin | 7 | |
| Aminoglycosides | 4 | 1 |
| Gentamicin | 4 | |
| Otherb | 43 | 16 |
| Antimicrobial Therapy (12 wks Before Stool Collection) . | n . | %a . |
|---|---|---|
| Any antimicrobial therapy | 200 | 76 |
| Fluoroquinolones | 78 | 30 |
| Ciprofloxacin | 51 | |
| Levofloxacin | 20 | |
| Moxifloxacin | 10 | |
| β-lactam/β-lactamase inhibitor combinations | 68 | 26 |
| Piperacillin-tazobactam | 45 | |
| Amoxicillin/clavulanic acid | 25 | |
| Ampicillin/sulbactam | 6 | |
| Glycopeptide | 55 | 21 |
| Vancomycin (IV) | 55 | |
| 3rd- and 4th-generation cephalosporins | 52 | 20 |
| Ceftriaxone | 39 | |
| Cefepime | 14 | |
| Cefpodoxime | 3 | |
| Ceftazidime | 1 | |
| Ceftizoxime | 1 | |
| 1st- and 2nd-generation cephalosporins | 33 | 13 |
| Cephalexin | 21 | |
| Cefazolin | 15 | |
| Cefotetan | 1 | |
| Macrolides | 23 | 9 |
| Azithromycin | 21 | |
| Erythromycin | 2 | |
| Clarithromycin | 1 | |
| Sulfa | 17 | 6 |
| Trimethoprim-sulfamethoxazole | 17 | |
| Carbapenems | 14 | 5 |
| Ertapenem | 7 | |
| Meropenem | 5 | |
| Imipenem | 2 | |
| Lincosamide | 13 | 5 |
| Clindamycin | 13 | |
| Penicillins | 9 | 3 |
| Amoxicillin | 7 | |
| Aminoglycosides | 4 | 1 |
| Gentamicin | 4 | |
| Otherb | 43 | 16 |
Abbreviations: CDI, Clostridium difficile infection; IV, intravenous; NHO, nursing home onset.
a Percentage of all cases with a full medical record review, n = 262.
b Other antibiotics include the following: metronidazole, doxycycline, linezolid, nitrofurantoin, daptomycin.
Among the 200 (76%) NHO-CDI cases discharged from a hospital in the 12 weeks prior to C difficile positive stool collection, 48% were known to have been hospitalized in the 2 weeks prior and 75% were known to have been hospitalized in the month prior (57% of all NHO-CDI cases; Figure 1). Few cases (<2%) had severe outcomes such as colectomy, pseudomembranous colitis, toxic megacolon, or ileus.
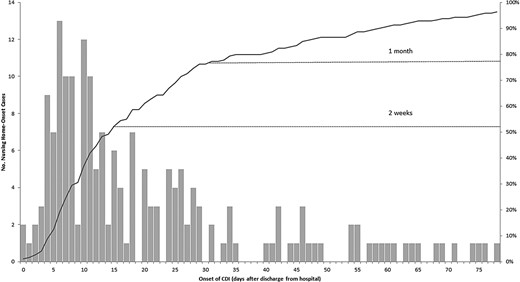
Number of days from hospital discharge to Clostridium difficile infection (CDI) onset among cases with hospitalization in 12 weeks prior to C difficile positive stool collection date (n = 200)*. *Figure does not include 14 patients who did not have a hospitalization date available.
Predictors of Increased Nursing Home-Onset Clostridium difficile Infection Incidence
Of candidate variables included in the generalized linear mixed model, only age was significantly associated with increased NHO-CDI incidence (P < .0001). Nucleic acid amplification test usage did not meet the stay criterion for inclusion in the model (P = .80), likely due to high NAAT usage across EIP sites. However, NAAT usage was retained in the final model for consistency with previously used methodology, and the 74% NAAT usage found across EIP sites was used to generate the national estimates. Controlling for NAAT usage, NHO-CDI incidence was more than 100-fold higher (95% CI, 91- to 134-fold) in persons 85 years and older compared with persons between 1 and 84 years of age.
Crude NHO-CDI incidence varied across the 10 EIP sites, with the lowest incidence in Site I (5.8 per 100 000 population) and highest in Site G (61.1 per 100 000 population) (Table 3). After adjusting for age and NAAT usage, the differences across sites did not change substantially; the sites with the highest and lowest incidences remained the same (7.4 cases per 100 000 for Site I; 66.1 cases per 100 000 population for Site G).
| Site . | NHO CDI Cases (N) . | Crude Rate, per 100 000 Personsa . | Adjusted Rate, per 100 000 Personsa,b . |
|---|---|---|---|
| Site A | 478 | 56.0 | 57.4 |
| (95% CI, 55.2–59.7) | |||
| Site Bc | 758 | 30.3 | 28.6 |
| (95% CI, 556–959) | (95% CI, 22.3–38.4) | (95% CI, 18.1–38.3) | |
| Site C | 219 | 26.7 | 28.6 |
| (95% CI, 27.8–29.3) | |||
| Site Dc | 637 | 16.9 | 15.7 |
| (95% CI, 430–843.8) | (95% CI, 11.4–22.4) | (95% CI, 9.6–21.7) | |
| Site E | 475 | 57.2 | 57.9 |
| (95% CI, 55.7–60.1) | |||
| Site F | 21 | 8.6 | 10.2 |
| (95% CI, 7.9–12.4) | |||
| Site G | 406 | 61.1 | 66.1 |
| (95% CI, 58.4–73.8) | |||
| Site H | 345 | 46.7 | 51.1 |
| (95% CI, 49.7–52.5) | |||
| Site I | 13 | 5.8 | 7.4 |
| (95% CI, 4.3–10.4) | |||
| Site J | 151 | 23.6 | 23.5 |
| (95% CI, 22.8–24.2) | |||
| Total | 3503 | 31.0 | 31.2 |
| (95% CI, 3095–3911) | (95% CI, 27.4–34.7) | (95% CI, 25.8–36.5) |
| Site . | NHO CDI Cases (N) . | Crude Rate, per 100 000 Personsa . | Adjusted Rate, per 100 000 Personsa,b . |
|---|---|---|---|
| Site A | 478 | 56.0 | 57.4 |
| (95% CI, 55.2–59.7) | |||
| Site Bc | 758 | 30.3 | 28.6 |
| (95% CI, 556–959) | (95% CI, 22.3–38.4) | (95% CI, 18.1–38.3) | |
| Site C | 219 | 26.7 | 28.6 |
| (95% CI, 27.8–29.3) | |||
| Site Dc | 637 | 16.9 | 15.7 |
| (95% CI, 430–843.8) | (95% CI, 11.4–22.4) | (95% CI, 9.6–21.7) | |
| Site E | 475 | 57.2 | 57.9 |
| (95% CI, 55.7–60.1) | |||
| Site F | 21 | 8.6 | 10.2 |
| (95% CI, 7.9–12.4) | |||
| Site G | 406 | 61.1 | 66.1 |
| (95% CI, 58.4–73.8) | |||
| Site H | 345 | 46.7 | 51.1 |
| (95% CI, 49.7–52.5) | |||
| Site I | 13 | 5.8 | 7.4 |
| (95% CI, 4.3–10.4) | |||
| Site J | 151 | 23.6 | 23.5 |
| (95% CI, 22.8–24.2) | |||
| Total | 3503 | 31.0 | 31.2 |
| (95% CI, 3095–3911) | (95% CI, 27.4–34.7) | (95% CI, 25.8–36.5) |
Abbreviations: CDI, Clostridium difficile infection; CI, confidence interval; NAAT, nucleic acid amplification test; NHO, nursing home onset.
a Population estimates from 2012 US Census.
b Adjusted for age and diagnostic test (ie, NAAT usage).
c Site did stratified sampling of cases based on age and sex; point estimate and 95% CI are presented for each surveillance site.
| Site . | NHO CDI Cases (N) . | Crude Rate, per 100 000 Personsa . | Adjusted Rate, per 100 000 Personsa,b . |
|---|---|---|---|
| Site A | 478 | 56.0 | 57.4 |
| (95% CI, 55.2–59.7) | |||
| Site Bc | 758 | 30.3 | 28.6 |
| (95% CI, 556–959) | (95% CI, 22.3–38.4) | (95% CI, 18.1–38.3) | |
| Site C | 219 | 26.7 | 28.6 |
| (95% CI, 27.8–29.3) | |||
| Site Dc | 637 | 16.9 | 15.7 |
| (95% CI, 430–843.8) | (95% CI, 11.4–22.4) | (95% CI, 9.6–21.7) | |
| Site E | 475 | 57.2 | 57.9 |
| (95% CI, 55.7–60.1) | |||
| Site F | 21 | 8.6 | 10.2 |
| (95% CI, 7.9–12.4) | |||
| Site G | 406 | 61.1 | 66.1 |
| (95% CI, 58.4–73.8) | |||
| Site H | 345 | 46.7 | 51.1 |
| (95% CI, 49.7–52.5) | |||
| Site I | 13 | 5.8 | 7.4 |
| (95% CI, 4.3–10.4) | |||
| Site J | 151 | 23.6 | 23.5 |
| (95% CI, 22.8–24.2) | |||
| Total | 3503 | 31.0 | 31.2 |
| (95% CI, 3095–3911) | (95% CI, 27.4–34.7) | (95% CI, 25.8–36.5) |
| Site . | NHO CDI Cases (N) . | Crude Rate, per 100 000 Personsa . | Adjusted Rate, per 100 000 Personsa,b . |
|---|---|---|---|
| Site A | 478 | 56.0 | 57.4 |
| (95% CI, 55.2–59.7) | |||
| Site Bc | 758 | 30.3 | 28.6 |
| (95% CI, 556–959) | (95% CI, 22.3–38.4) | (95% CI, 18.1–38.3) | |
| Site C | 219 | 26.7 | 28.6 |
| (95% CI, 27.8–29.3) | |||
| Site Dc | 637 | 16.9 | 15.7 |
| (95% CI, 430–843.8) | (95% CI, 11.4–22.4) | (95% CI, 9.6–21.7) | |
| Site E | 475 | 57.2 | 57.9 |
| (95% CI, 55.7–60.1) | |||
| Site F | 21 | 8.6 | 10.2 |
| (95% CI, 7.9–12.4) | |||
| Site G | 406 | 61.1 | 66.1 |
| (95% CI, 58.4–73.8) | |||
| Site H | 345 | 46.7 | 51.1 |
| (95% CI, 49.7–52.5) | |||
| Site I | 13 | 5.8 | 7.4 |
| (95% CI, 4.3–10.4) | |||
| Site J | 151 | 23.6 | 23.5 |
| (95% CI, 22.8–24.2) | |||
| Total | 3503 | 31.0 | 31.2 |
| (95% CI, 3095–3911) | (95% CI, 27.4–34.7) | (95% CI, 25.8–36.5) |
Abbreviations: CDI, Clostridium difficile infection; CI, confidence interval; NAAT, nucleic acid amplification test; NHO, nursing home onset.
a Population estimates from 2012 US Census.
b Adjusted for age and diagnostic test (ie, NAAT usage).
c Site did stratified sampling of cases based on age and sex; point estimate and 95% CI are presented for each surveillance site.
National Estimates
After adjusting for age group and a presumed NAAT usage of 74%, we estimated that 112 800 cases of NHO CDI (95% CI, 93 400–131 800) occurred in the United States in 2012 (Table 4). Of the 112 800 NHO-CDI cases, we estimated that 31 400 (28%) were hospitalized within 7 days after a positive specimen (95% CI, 25 500–37 300), 20 900 (19%) had an initial recurrence between 14 and 60 days of the last positive specimen (95% CI, 14 600–27 100), and 8700 (8%) died within 30 days (95% CI, 6600–10 700). Incidence, hospitalization, recurrence, and death rates were highest in persons 85 years or older.
National Estimates of NHO-CDI Incidence, Hospitalization, Recurrence, and Death—United States, 2012
| Characteristic . | Estimated Incidencea . | Estimated Hospitalizationb . | Estimated Recurrenceb . | Estimated Deathsb . | ||||
|---|---|---|---|---|---|---|---|---|
| Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | |
| Sex | ||||||||
| Male | 47 000 (38 800–55 200) | 30.8 (25.5–36.2) | 12 400 (7200–17 700) | 8.1 (4.7–11.6) | 7400 (2100–12 800) | 4.9 (1.4–8.4) | 4400 (1900–6900) | 2.9 (1.3–4.5) |
| Female | 65 700 (54 600–76 500) | 41.7 (34.7–48.6) | 19 000 (9300–28 600) | 12.1 (5.9–18.2) | 13 500 (5300–21 700) | 8.6 (3.4–13.8) | 4 200 (1300–7200) | 2.7 (.8–4.6) |
| Age Group | ||||||||
| <65 | 15 200 (11 800–18 200) | 5.7 (4.4–6.8) | 5000 (2700–7300) | 1.9 (1–2.7) | 1500 (0–3100) | .6 (0–1.2) | 200 (0–400) | .1 (0–.2) |
| 65–84 | 57 700 (48 700–66 700) | 154.8 (130.8–178.9) | 15 500 (8200–22 800) | 41.6 (22–61.2) | 10 400 (3200–17 600) | 27.9 (8.6–47.2) | 4800 (700–8900) | 12.9 (1.9–23.9) |
| ≥85 | 39 900 (32 900–47 000) | 677.9 (558.0–797.8) | 10 900 (5600–16 100) | 185.1 (95.1–273.5) | 9000 (3700–14 300) | 152.9 (62.9–242.9) | 3700 (700–6700) | 62.9 (11.9–113.8) |
| Race | ||||||||
| White | 99 200 (83 900–114 500) | 41.0 (34.7–47.4) | 26 100 (14 100–38 100) | 10.8 (5.8–15.8) | 18 600 (6900–30 300) | 7.7 (2.9–12.5) | 7500 (2900–12 100) | 3.1 (1.2–5) |
| Other | 13 600 (9500–17 300) | 19.9 (13.9–25.3) | 5300 (1200–9400) | 7.8 (1.8–13.8) | 2300 (0–5100) | 3.4 (0–7.5) | 1100 (100–2200) | 1.6 (.2–3.2) |
| Total | 112 800 (93 400–131 800) | 36.4 (30.1–42.5) | 31 400 (25 500–37 300) | 10.1 (8.2–12.0) | 20 900 (14 600–27 100) | 6.7 (4.7–8.7) | 8700 (6600–10 700) | 2.8 (2.1–3.5) |
| Characteristic . | Estimated Incidencea . | Estimated Hospitalizationb . | Estimated Recurrenceb . | Estimated Deathsb . | ||||
|---|---|---|---|---|---|---|---|---|
| Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | |
| Sex | ||||||||
| Male | 47 000 (38 800–55 200) | 30.8 (25.5–36.2) | 12 400 (7200–17 700) | 8.1 (4.7–11.6) | 7400 (2100–12 800) | 4.9 (1.4–8.4) | 4400 (1900–6900) | 2.9 (1.3–4.5) |
| Female | 65 700 (54 600–76 500) | 41.7 (34.7–48.6) | 19 000 (9300–28 600) | 12.1 (5.9–18.2) | 13 500 (5300–21 700) | 8.6 (3.4–13.8) | 4 200 (1300–7200) | 2.7 (.8–4.6) |
| Age Group | ||||||||
| <65 | 15 200 (11 800–18 200) | 5.7 (4.4–6.8) | 5000 (2700–7300) | 1.9 (1–2.7) | 1500 (0–3100) | .6 (0–1.2) | 200 (0–400) | .1 (0–.2) |
| 65–84 | 57 700 (48 700–66 700) | 154.8 (130.8–178.9) | 15 500 (8200–22 800) | 41.6 (22–61.2) | 10 400 (3200–17 600) | 27.9 (8.6–47.2) | 4800 (700–8900) | 12.9 (1.9–23.9) |
| ≥85 | 39 900 (32 900–47 000) | 677.9 (558.0–797.8) | 10 900 (5600–16 100) | 185.1 (95.1–273.5) | 9000 (3700–14 300) | 152.9 (62.9–242.9) | 3700 (700–6700) | 62.9 (11.9–113.8) |
| Race | ||||||||
| White | 99 200 (83 900–114 500) | 41.0 (34.7–47.4) | 26 100 (14 100–38 100) | 10.8 (5.8–15.8) | 18 600 (6900–30 300) | 7.7 (2.9–12.5) | 7500 (2900–12 100) | 3.1 (1.2–5) |
| Other | 13 600 (9500–17 300) | 19.9 (13.9–25.3) | 5300 (1200–9400) | 7.8 (1.8–13.8) | 2300 (0–5100) | 3.4 (0–7.5) | 1100 (100–2200) | 1.6 (.2–3.2) |
| Total | 112 800 (93 400–131 800) | 36.4 (30.1–42.5) | 31 400 (25 500–37 300) | 10.1 (8.2–12.0) | 20 900 (14 600–27 100) | 6.7 (4.7–8.7) | 8700 (6600–10 700) | 2.8 (2.1–3.5) |
Abbreviations: CDI, Clostridium difficile infection; CI, confidence interval; NAAT, nucleic acid amplification test; NHO, nursing home onset.
a Estimates calculated using NHO-specific generalized mixed model with negative binomial distribution, controlling for age and diagnostic test (ie, NAAT usage).
b Estimates calculated using imputation and domain analysis based on distribution of outcomes for each age, sex, and epidemiologic class category.
c Rounded to the nearest hundred.
d Rate per 100 000 population, rounded to the nearest 10th.
National Estimates of NHO-CDI Incidence, Hospitalization, Recurrence, and Death—United States, 2012
| Characteristic . | Estimated Incidencea . | Estimated Hospitalizationb . | Estimated Recurrenceb . | Estimated Deathsb . | ||||
|---|---|---|---|---|---|---|---|---|
| Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | |
| Sex | ||||||||
| Male | 47 000 (38 800–55 200) | 30.8 (25.5–36.2) | 12 400 (7200–17 700) | 8.1 (4.7–11.6) | 7400 (2100–12 800) | 4.9 (1.4–8.4) | 4400 (1900–6900) | 2.9 (1.3–4.5) |
| Female | 65 700 (54 600–76 500) | 41.7 (34.7–48.6) | 19 000 (9300–28 600) | 12.1 (5.9–18.2) | 13 500 (5300–21 700) | 8.6 (3.4–13.8) | 4 200 (1300–7200) | 2.7 (.8–4.6) |
| Age Group | ||||||||
| <65 | 15 200 (11 800–18 200) | 5.7 (4.4–6.8) | 5000 (2700–7300) | 1.9 (1–2.7) | 1500 (0–3100) | .6 (0–1.2) | 200 (0–400) | .1 (0–.2) |
| 65–84 | 57 700 (48 700–66 700) | 154.8 (130.8–178.9) | 15 500 (8200–22 800) | 41.6 (22–61.2) | 10 400 (3200–17 600) | 27.9 (8.6–47.2) | 4800 (700–8900) | 12.9 (1.9–23.9) |
| ≥85 | 39 900 (32 900–47 000) | 677.9 (558.0–797.8) | 10 900 (5600–16 100) | 185.1 (95.1–273.5) | 9000 (3700–14 300) | 152.9 (62.9–242.9) | 3700 (700–6700) | 62.9 (11.9–113.8) |
| Race | ||||||||
| White | 99 200 (83 900–114 500) | 41.0 (34.7–47.4) | 26 100 (14 100–38 100) | 10.8 (5.8–15.8) | 18 600 (6900–30 300) | 7.7 (2.9–12.5) | 7500 (2900–12 100) | 3.1 (1.2–5) |
| Other | 13 600 (9500–17 300) | 19.9 (13.9–25.3) | 5300 (1200–9400) | 7.8 (1.8–13.8) | 2300 (0–5100) | 3.4 (0–7.5) | 1100 (100–2200) | 1.6 (.2–3.2) |
| Total | 112 800 (93 400–131 800) | 36.4 (30.1–42.5) | 31 400 (25 500–37 300) | 10.1 (8.2–12.0) | 20 900 (14 600–27 100) | 6.7 (4.7–8.7) | 8700 (6600–10 700) | 2.8 (2.1–3.5) |
| Characteristic . | Estimated Incidencea . | Estimated Hospitalizationb . | Estimated Recurrenceb . | Estimated Deathsb . | ||||
|---|---|---|---|---|---|---|---|---|
| Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | Burden Estimatec (95% CI) . | Rated (95% CI) . | |
| Sex | ||||||||
| Male | 47 000 (38 800–55 200) | 30.8 (25.5–36.2) | 12 400 (7200–17 700) | 8.1 (4.7–11.6) | 7400 (2100–12 800) | 4.9 (1.4–8.4) | 4400 (1900–6900) | 2.9 (1.3–4.5) |
| Female | 65 700 (54 600–76 500) | 41.7 (34.7–48.6) | 19 000 (9300–28 600) | 12.1 (5.9–18.2) | 13 500 (5300–21 700) | 8.6 (3.4–13.8) | 4 200 (1300–7200) | 2.7 (.8–4.6) |
| Age Group | ||||||||
| <65 | 15 200 (11 800–18 200) | 5.7 (4.4–6.8) | 5000 (2700–7300) | 1.9 (1–2.7) | 1500 (0–3100) | .6 (0–1.2) | 200 (0–400) | .1 (0–.2) |
| 65–84 | 57 700 (48 700–66 700) | 154.8 (130.8–178.9) | 15 500 (8200–22 800) | 41.6 (22–61.2) | 10 400 (3200–17 600) | 27.9 (8.6–47.2) | 4800 (700–8900) | 12.9 (1.9–23.9) |
| ≥85 | 39 900 (32 900–47 000) | 677.9 (558.0–797.8) | 10 900 (5600–16 100) | 185.1 (95.1–273.5) | 9000 (3700–14 300) | 152.9 (62.9–242.9) | 3700 (700–6700) | 62.9 (11.9–113.8) |
| Race | ||||||||
| White | 99 200 (83 900–114 500) | 41.0 (34.7–47.4) | 26 100 (14 100–38 100) | 10.8 (5.8–15.8) | 18 600 (6900–30 300) | 7.7 (2.9–12.5) | 7500 (2900–12 100) | 3.1 (1.2–5) |
| Other | 13 600 (9500–17 300) | 19.9 (13.9–25.3) | 5300 (1200–9400) | 7.8 (1.8–13.8) | 2300 (0–5100) | 3.4 (0–7.5) | 1100 (100–2200) | 1.6 (.2–3.2) |
| Total | 112 800 (93 400–131 800) | 36.4 (30.1–42.5) | 31 400 (25 500–37 300) | 10.1 (8.2–12.0) | 20 900 (14 600–27 100) | 6.7 (4.7–8.7) | 8700 (6600–10 700) | 2.8 (2.1–3.5) |
Abbreviations: CDI, Clostridium difficile infection; CI, confidence interval; NAAT, nucleic acid amplification test; NHO, nursing home onset.
a Estimates calculated using NHO-specific generalized mixed model with negative binomial distribution, controlling for age and diagnostic test (ie, NAAT usage).
b Estimates calculated using imputation and domain analysis based on distribution of outcomes for each age, sex, and epidemiologic class category.
c Rounded to the nearest hundred.
d Rate per 100 000 population, rounded to the nearest 10th.
Clostridium difficile Strain Types
Of the 262 NHO-CDI cases with full medical record review, 76 (29%) had isolates that underwent molecular characterization (Table 5). Ribotype 027 was the most prevalent (30% of isolates). Other strain types identified include ribotypes 002 (11%), 106 (7%), and 078 (7%); 26 other ribotypes were represented in the remaining 45% of isolates typed. All EIP sites submitted isolates for molecular characterization; however, 80% of samples came from 5 of the 10 EIP sites. Therefore, geographic variation in ribotype could not be assessed.
| Ribotype . | No. of Cases (n = 76) . | % . |
|---|---|---|
| 027 | 23 | 30 |
| 002 | 8 | 11 |
| 106 | 5 | 7 |
| 078 | 5 | 7 |
| 053 | 3 | 4 |
| 012 | 2 | 3 |
| 014 | 2 | 3 |
| 017 | 2 | 3 |
| 020 | 2 | 3 |
| 046 | 2 | 3 |
| 054 | 2 | 3 |
| 001/072 | 1 | 1 |
| 015 | 1 | 1 |
| 039 | 1 | 1 |
| 043 | 1 | 1 |
| 076 | 1 | 1 |
| 097 | 1 | 1 |
| 103 | 1 | 1 |
| 153 | 1 | 1 |
| 356 | 1 | 1 |
| 570 | 1 | 1 |
| 374 | 1 | 1 |
| Others | 9 | 11 |
| Ribotype . | No. of Cases (n = 76) . | % . |
|---|---|---|
| 027 | 23 | 30 |
| 002 | 8 | 11 |
| 106 | 5 | 7 |
| 078 | 5 | 7 |
| 053 | 3 | 4 |
| 012 | 2 | 3 |
| 014 | 2 | 3 |
| 017 | 2 | 3 |
| 020 | 2 | 3 |
| 046 | 2 | 3 |
| 054 | 2 | 3 |
| 001/072 | 1 | 1 |
| 015 | 1 | 1 |
| 039 | 1 | 1 |
| 043 | 1 | 1 |
| 076 | 1 | 1 |
| 097 | 1 | 1 |
| 103 | 1 | 1 |
| 153 | 1 | 1 |
| 356 | 1 | 1 |
| 570 | 1 | 1 |
| 374 | 1 | 1 |
| Others | 9 | 11 |
| Ribotype . | No. of Cases (n = 76) . | % . |
|---|---|---|
| 027 | 23 | 30 |
| 002 | 8 | 11 |
| 106 | 5 | 7 |
| 078 | 5 | 7 |
| 053 | 3 | 4 |
| 012 | 2 | 3 |
| 014 | 2 | 3 |
| 017 | 2 | 3 |
| 020 | 2 | 3 |
| 046 | 2 | 3 |
| 054 | 2 | 3 |
| 001/072 | 1 | 1 |
| 015 | 1 | 1 |
| 039 | 1 | 1 |
| 043 | 1 | 1 |
| 076 | 1 | 1 |
| 097 | 1 | 1 |
| 103 | 1 | 1 |
| 153 | 1 | 1 |
| 356 | 1 | 1 |
| 570 | 1 | 1 |
| 374 | 1 | 1 |
| Others | 9 | 11 |
| Ribotype . | No. of Cases (n = 76) . | % . |
|---|---|---|
| 027 | 23 | 30 |
| 002 | 8 | 11 |
| 106 | 5 | 7 |
| 078 | 5 | 7 |
| 053 | 3 | 4 |
| 012 | 2 | 3 |
| 014 | 2 | 3 |
| 017 | 2 | 3 |
| 020 | 2 | 3 |
| 046 | 2 | 3 |
| 054 | 2 | 3 |
| 001/072 | 1 | 1 |
| 015 | 1 | 1 |
| 039 | 1 | 1 |
| 043 | 1 | 1 |
| 076 | 1 | 1 |
| 097 | 1 | 1 |
| 103 | 1 | 1 |
| 153 | 1 | 1 |
| 356 | 1 | 1 |
| 570 | 1 | 1 |
| 374 | 1 | 1 |
| Others | 9 | 11 |
DISCUSSION
We estimated that C difficile caused approximately 113 000 infections with onset in nursing homes in the United States in 2012, representing approximately one quarter of all US CDI cases. The true burden of CDI in nursing homes is likely higher because we do not account for residents entering nursing homes with CDI onset in different settings [21]. This number has the potential to increase as the population ages and the number of nursing home residents in the United States grows [22]. Although approximately 32 000 NHO-CDI cases were hospitalized within 7 days after infection, a large proportion (73%) did not require hospitalization. This demonstrates the importance of accounting for CDI with onset in nursing homes for guiding prevention efforts and monitoring progress; relying solely on hospital-based surveillance underestimates the CDI burden across healthcare settings.
Antibiotic use, the most important risk factor for C difficile susceptibility, was common among NHO-CDI cases. Because antibiotics can alter the intestinal microbiota for 3 months or longer and predispose patients to infection with C difficile [7], prior antibiotic use in an acute care setting can increase patients' susceptibility during the nursing home stay. We found that the majority (76%) of NHO-CDI cases had recently been discharged from a hospital prior to onset of illness, and 75% of these residents had antibiotic exposure. The incubation period for C difficile is uncertain, but it is likely short (median of 2–3 days, [23, 24]), suggesting that the temporal clustering of cases in the 2–4 weeks after discharge from hospital to nursing home reflects this increased susceptibility to CDI among those with recent hospitalization. Similar observations have been reported in other studies [12, 13]. It is possible that some NHO-CDI cases may have acquired C difficile while in the acute care setting, given that 36% had a positive C difficile-positive stool within 2 weeks after discharge, particularly if early symptoms were not recognized and diagnosed. These findings suggest decreasing NHO CDI will require antibiotic stewardship in acute and long-term care settings to limit the number of residents susceptible to CDI combined with improved infection prevention measures in both settings to reduce exposure C difficile spores.
We found that NHO-CDI incidence varied by geographic region, and this variation persisted after adjusting for age and type of diagnostic test. Factors associated with high incidence that were not accounted for in our analysis, such as geographic variability in antimicrobial prescribing, ribotype distribution, infection control practices, or physician testing practices (eg, empiric CDI therapy with no stool testing), could be contributing to the variability of CDI rates across EIP site. Nursing home-onset CDI incidence was correlated with overall healthcare-associated CDI incidence by surveillance site; the 4 geographic regions with the highest healthcare-associated CDI also had the highest NHO-CDI rates [4]. Further research is needed to better understand contributors to this regional variation.
The distribution of ribotypes in NHO-CDI cases has not previously been reported. The epidemic ribotype 027, which has been associated with more severe infection and was reported as a cause of many hospital outbreaks in the United States and Canada [25], was most prevalent among the NHO-CDI isolates tested in our study; other ribotypes included 002, 106, and 078. These findings differ from those reported from a recent hospital-based survey in England where ribotype 027 has been decreasing, along with CDI rates, such that it is no longer among the most prevalent strains [26, 27]. In another European hospital-based survey, ribotypes 014, 001, and 078 were most common, with 027 accounting for fewer than 5% of isolates [28]. Ribotype 078, identified in our study and the European hospital survey, is the predominant C difficile strain identified in food animals in the United States [28, 29]. However, these findings are based on a small percentage of isolates (5%) tested in our study from a convenience sample of laboratories and may not be representative of laboratory samples within EIP or nationally.
There are several limitations to this study. First, although the data are drawn from surveillance covering a large population and including demographically diverse geographic regions, EIP sites were not selected to be nationally representative. To produce estimates reflecting the distribution of the population in the United States, we evaluated regional differences in age, sex, race, nursing home and hospital utilization, and diagnostic testing, and then we adjusted for the variables whose association with incidence varied by region. Second, we present NHO-CDI incidence based on total population in the surveillance area, not the population in nursing homes, which would require additional facility-level information (ie, resident-days). Third, our surveillance definition relies on C difficile-positive testing. Therefore, it is possible that we may have underestimated CDI rates if physicians did not suspect CDI and order testing in symptomatic patients or if patients were treated empirically without diagnostic testing [30–32]. Alternatively, we may have overestimated CDI rates if some positive testes were obtained from patients without clinically relevant diarrhea (representing colonization) [31, 33, 34]. In addition, the surveillance data only capture initial NHO-CDI recurrences; some patients will have multiple recurrences that are not included in these estimates. Fourth, clinical and outcome data were only evaluated from a subset of cases, and we relied on data available in medical record; for example, we suspect that a greater number of NHO-CDI cases had diarrhea, but this may not have been documented. Although cases for medical record review were systematically selected to increase the representativeness of these data, the stratified sampling of cases in Colorado and Georgia was based on age categories that differed from the nursing home-specific age groups used in our analysis, which may have overrepresented the fact that younger patients had an impact on the death and recurrence estimates. Fifth, laboratory testing method was not available at the case-level; therefore, it was not possible to stratify results by test type (ie, immunoassay, NAAT). Lastly, we estimated hospitalization and death subsequent to onset of CDI, but we were not able to determine the proportions of hospitalization or deaths attributable to CDI.
The estimated number of NHO CDI in 2012 (112 800; 95% CI, 93 400–131 800) is 3% lower than was reported in 2011 [35] after adjusting for increased adoption of NAAT usage (116 000; 95% CI, 104 600–127 800), a more sensitive laboratory diagnostic method, which increased from 52% to 74% between 2011 and 2012 [4].
CONCLUSIONS
This study used data from multiple geographic locations to provide a national estimate of NHO-CDI burden. By characterizing the magnitude of the issue and increasing our understanding of risk factors, this work—along with continued surveillance—can help guide and evaluate the effectiveness of CDI prevention policies. Research is needed to further understand how specific prevention efforts in both acute and long-term care settings, including reductions in unnecessary antibiotic use and improvements in infection prevention programs, can lead to decreases in CDI in nursing homes.
Acknowledgments
We acknowledge the following contributors: Erin Garcia, Erin Parker, Joelle Nadle, and Gretchen Rothrock (California Emerging Infections Program); Carol Lyons (Connecticut Emerging Infections Program); Wendy Bamberg (Colorado Emerging Infections Program); Olivia Almendares, Wendy Baughman, Leigh Ann Clark, Andrew Revis, and Zirka Smith (Georgia Emerging Infections Program); Lucy Wilson (Maryland Emerging Infections Program); Ruth Lynfield (Minnesota Emerging Infections Program); Nicole Kenslow (New Mexico Emerging Infections Program); Rebecca Tsay and Deborah Nelson (New York Emerging Infections Program); Valerie Ocampo (Oregon Emerging Infections Program); Samir Hanna, Amanda Ingram, Brenda Rue, and Corrine Davis (Tennessee Emerging Infections Program); Susan Sambol and Laurica Petrella (Hines VA Hospital); Brandi Limbago, James K. Rasheed, and Ashley Paulick (Centers for Disease Control and Prevention).
Disclaimer. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Financial support. This work was funded by the Emerging Infections Program Cooperative Agreement between the 10 EIP sites and the Centers for Disease Control and Prevention.
Potential conflicts of interest. Dr Gerding is a board member of Merck, Rebiotix, Summit, and Actelion, and consults for Sanofi Pasteur, DaVolterra, Pfizer, and Cubist, all of which perform research on potential C difficile products. D. N. G. is a consultant for and has patents licensed to Shire. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
Author notes
Presented in part: 2014 IDWeek Meeting, Philadelphia, PA.
- antibiotics
- medical records
- clostridium difficile infections
- exposure
- feces
- internship and residency
- laboratory techniques and procedures
- long-term care
- nursing homes
- outpatients
- morbidity
- mortality
- specimen collection
- infection prophylaxis
- acute care
- surveillance, medical
- medical residencies
- hospital admission
- clostridium difficile
- nursing home resident
- patient-focused outcomes
- national health service (uk)





Comments