-
PDF
- Split View
-
Views
-
Cite
Cite
Simon van der Pol, Maartje S Jacobs, Karina Meijer, Margriet G Piersma-Wichers, Robert G Tieleman, Maarten J Postma, Marinus van Hulst, Perioperative bridging of vitamin K antagonist treatment in patients with atrial fibrillation: only a very small group of patients benefits, EP Europace, Volume 21, Issue 5, May 2019, Pages 716–723, https://doi.org/10.1093/europace/euy308
Close - Share Icon Share
Abstract
Bridging anticoagulation in atrial fibrillation (AF) patients who need to interrupt vitamin K antagonists for procedures is a clinical dilemma. Currently, guidelines recommend clinicians to take the stroke and bleeding risk into consideration, but no clear thresholds are advised. To aid clinical decision making, we aimed to develop a model in which periprocedural bridging therapy is compared with withholding anticoagulation in AF patients, for several bleeding and stroke risk groups.
A model was developed to simulate both a bridge and a non-bridge cohort, using simulated international normalized ratio (INR) values for patients on warfarin, acenocoumarol, and phenprocoumon. For both clinical strategies, stroke and bleeding risks were included and outcomes were stratified by CHA2DS2-VASc or CHADS2 and HAS-BLED groups. Quality-adjusted life expectancy was the main outcome considered. Our analyses show bridging to only be beneficial for patients with HAS-BLED scores equal or lower to 2 and with CHA2DS2-VASc scores of 6 or higher. For patients using acenocoumarol bridging may be beneficial starting at a CHA2DS2-VASc score of 7. Post-procedural time to therapeutic INR has a significant influence on the results: no significant benefit of bridging was found for patients reaching therapeutic INR values within 5 days.
When deciding whether to bridge anticoagulation, clinicians should consider the patient’s individual stroke and bleeding risk, while also considering the patient’s post-procedural INR management. In practice, only a small subset of patients is expected to benefit from bridging anticoagulation treatment.
The Bridge trial by Douketis et al. concluded that bridging warfarin periprocedurally with dalteparin was not beneficial. However, there has been much debate regarding the potential advantages for specific patient groups, not included in the beforementioned trial, and guidelines still advice the use of bridging anticoagulation for these groups.
We built a model that shows that for most atrial fibrillation patients, bridging anticoagulation is not likely to be beneficial, although small subgroups can be determined where bridging anticoagulation may yield better life expectancy outcomes.
Optimal international normalized ratio management, where therapeutic values are reached within 5 days, will improve life expectancy more than any benefit bridging of anticoagulants may have.
Introduction
Anticoagulant treatment reduces the risk of stroke in patients diagnosed with atrial fibrillation (AF).1 As they increase the risk of bleeding, anticoagulants have to be interrupted prior to a procedure if the risk of bleeding is considered high.2 Oral vitamin K antagonists (VKAs) are discontinued around 5 days prior to planned surgery; if the stroke risk is expected to be high, low-molecular-weight heparins (LMWHs), or unfractioned heparin can be administered to bridge this short ‘unprotected’ period, referred to as bridging anticoagulation.2 However, perioperative bridging is known to significantly increase the bleeding risk, enhancing discussion on the appropriateness of bridging.3 Notably, the recent BRIDGE trial (Perioperative Bridging Anticoagulation in Patients with Atrial Fibrillation) by Douketis et al.3 showed no added value of bridging therapy in AF patients. However, the BRIDGE trial included patients with a low average stroke risk (average CHADS2 score of 2.3 and 2.4, for the non-bridging and bridging arms, respectively) and might therefore have limited clinical validity.3
According to current guidelines, VKAs need to be interrupted if the procedural bleed risk or the patient bleed risk is increased and perioperative bridging anticoagulation should be considered if the annualized thrombotic risk is 5% or higher.2 These recommendations are mainly based on expert opinion: there is no clear clinical evidence to substantiate these claims.2 The CHA2DS2-VASc and CHADS2 scores can be used to determine the stroke risk. Bleeding risk mainly depends on the type of procedure, though bleeding risk will also vary per patient as expressed in their individual HAS-BLED score.2,4,5 A previous modelling study showed that perioperative anticoagulation is superior to non-bridging if a patient’s annual stroke rate exceeds 5.6%, or there is a less than 2.0% increase in bleeding risk caused by heparin.6 More recently, outcomes of bridging vs. non-bridging were simulated in a Monte Carlo simulation model, and it was concluded that patients at highest risk of ischaemic complications will benefit from bridging anticoagulation.7
We aimed to develop a model that compares perioperative VKA bridging to withholding anticoagulation for different stroke and bleeding risk subgroups considering different VKAs and procedures, resulting in straightforward clinical outcomes that can be used in medical decision making.
Methods
Model design
A Markov model (Figure 1) was developed to compare a bridge and a non-bridge cohort. The model starts with 1000 patients with two main stages being defined:
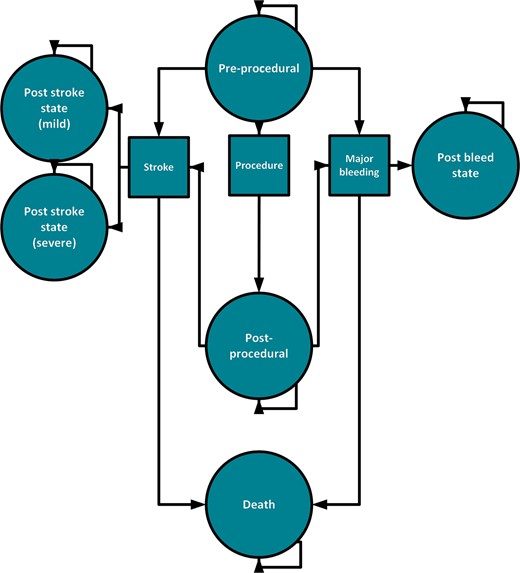
Markov model. The schematic representation of the Markov model used to simulate the perioperative period for atrial fibrillation patients on vitamin K antagonists. Circles represent health states, squares represent events, and arrows indicate transitions.
Pre-procedural stage: 5-day period before the procedure, since warfarin is usually interrupted 4–6 days prior to the procedure.2 Stroke and bleeding rates were based on AF population parameters.
Post-procedural stage: the 30-day follow-up period after the procedure, which is an often-used period for both bleeding and stroke in clinical studies.3 Stroke risk was based on either the CHADS2 or CHA2DS2-VASc population parameters, bleeding rates were derived from the BRIDGE trial.3–5
In line with the above, patients can undergo three events in the model:
Procedure: a surgical procedure, with intraprocedural events not being specifically included in the model, as the 24-h period around the procedure is assumed to have the same probabilities for specific events and complications as the pre-procedural period. All patients without a pre-procedural stroke or bleeding underwent surgery.
Stroke: an ischaemic stroke, stratified in mild (modified Rankin Scale 0–3), severe (4–5), and fatal (6). Stroke survivors entered the post-stroke state.
Major bleeding: as defined by the International Society on Thrombosis and Haemostasis and as used in the BRIDGE trial, including fatal bleeding.3,8 Patients surviving a bleeding event entered the post-bleeding state.
The model was build using R and several packages (see Supplementary material online, Table S1, for a complete list).9
Transition probabilities
Supplementary material online, Tables S2–S7 list all parameters that were used as model input. The stroke risk for both cohorts was simulated using international normalized ratio (INR) values and the odds ratios for stroke as reported in a trial, using a method previously described.7 We assumed non-bridging patients gradually moved from an INR value of 2.5 to 1.0 preoperatively and back to 2.5 post-operatively, using a normal logarithmic function. For the bridging cohort, a LMWH was administered during this period, up to 24 h prior to the procedure; post-operatively LMWH administration started 24 h after the procedure and was assumed discontinued when the INR reached 2.5. In the 48 h-period around the procedure, the INR was assumed to be 1.0, thus increasing the stroke risk.
The post-procedural period to reach an INR of 2.5 was assumed to vary between 5 and 15 days. Post-procedurally, the stroke risk was tripled when compared with the pre-procedural probabilities, based on the stroke rates of the BRIDGE trial.3,10 Since this parameter estimate was uncertain and not stratified for the CHADS2 or CHA2DS2-VASc subgroups, a wide beta-PERT distribution was applied in the probabilistic analysis. Regarding the bleeding risk, the bleeding rate reported in the BRIDGE study was used for the non-bridge cohort and the corresponding relative risk was applied to the bridge group.3 Low and high bleeding rates were differentiated using data from Omran et al.,3,11 assuming the populations to be comparable. We considered patients with a HAS-BLED score of 0–2 to have a low bleeding risk and a score of 3 or higher to have a high risk.11
Health outcomes and utilities
The clinical outcomes we looked at, stroke (mild and severe) and bleeding events, are not of an equal magnitude: stroke often has a permanent impact on the quality of life, bleeding events usually are restricted to short-term complications. To account for these differences, the declining exponential approximation of life expectancy was calculated to approximate the life expectancy, using the population parameters as reported by Statistics Netherlands and the AF incidence as reported in literature.12 The effect of the modified Rankin Scale score on the life expectancy was derived from Chiu et al.13 As a base-case, data for 75–80-year-old women was applied.
Calculated life expectancies were converted into Quality Adjusted Life Expectancies using utility values. For the stroke survivors, long-term utility values were used to differentiate the mild (modified Rankin Scale 0–3) and severe (4–5) groups. The impact on the quality of life of major bleeding was assumed to be negligible. Death was set to a quality of life of 0.
Simulation of international normalized ratio
The INR course after the procedure has been modelled using Eq. (2). For the base-case, all patients are assumed to reach an INR of 2.5 in 10 days, thus Eq. (2) is capped after this period. This is a conservative estimate, though not unrealistic, in clinical practice. The uncertainty of the INR trajectory was modelled by varying the variable q, with a mean of 0.092 [normally distributed, 95% confidence interval (CI) 0.069–0.18]. The impact of the INR trajectory was explored using separate scenarios where a post-operative therapeutic INR of 2.5 was reached post-operatively in 5, 10, or 15 days (fixed).
Sensitivity analyses
Random samples of the distribution for the model parameters were used in a Monte Carlo simulation consisting of 10 000 calculations. The results were recorded and used to calculate the mean and both the 2.5th and 97.5th percentile score, to approach the 95% CI of the mean. Results were considered statistically significant at the conventional cut-off at P = 0.05. As a base case warfarin was considered, being the most used VKA in Europe.14 Acenocoumarol and phenprocoumon were considered as alternatives, where the pre-procedural period was changed to 3 and 7 days, respectively, to account for the different half-lives of these VKAs.15
Results
In Figure 2, the stroke and bleeding rates are displayed for the base case. The rates of strokes ranged from less than 0.02% to almost 10% for the non-bridging group and less than 0.01% to almost 6% for the bridging group for the different CHA2DS2-VASc scores; bleeding rates varied from 0.03% to over 4% for low and high HAS-BLED scores for the non-bridging group and almost 1% to almost 10% for the bridging cohort. For the outcomes using the CHADS2 scores, see Supplementary material online, Figure S1.
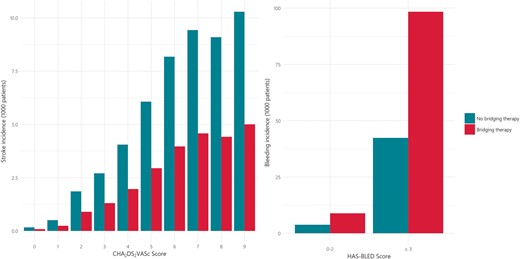
Stroke and bleeding outcomes in the simulation. Outcomes reported are for women of 75–80 years old. Left: stratified by CHA2DS2-VASc score as a percentage of the population. Right: stratified by HAS-BLED score as a percentage of the population. CHA2DS2-VASc, congestive heart failure, hypertension, age, diabetes, stroke, transient ischaemic attack or thromboembolism, vascular disease, age and sex; HAS-BLED, hypertension, abnormal renal and liver function, stroke, bleeding, labile INR, elderly, drugs/alcohol.
Figure 3 shows whether our simulation support bridging or not and whether the result was significant for the various age categories and both women and men, the results are stratified by CHA2DS2-VASc and HAS-BLED scores. As an example, for a female patient, aged 76, with a CHA2DS2-VASc score of 4 and a HAS-BLED score of 3 we do not expect a benefit if she is bridged. In general, the benefit of bridging was greater in younger patients and at higher CHA2DS2-VASc scores. For HAS-BLED scores of 3 and higher, no statistically significant benefit of bridging was found, regardless of the stroke risk. Figure 3 is based on the Monte Carlo simulation, which is displayed in more detail in the Supplementary material online, Figures S2 and S3; the equivalents using the CHADS2 stroke risk scores are displayed in Supplementary material online, Figures S4 and S5.
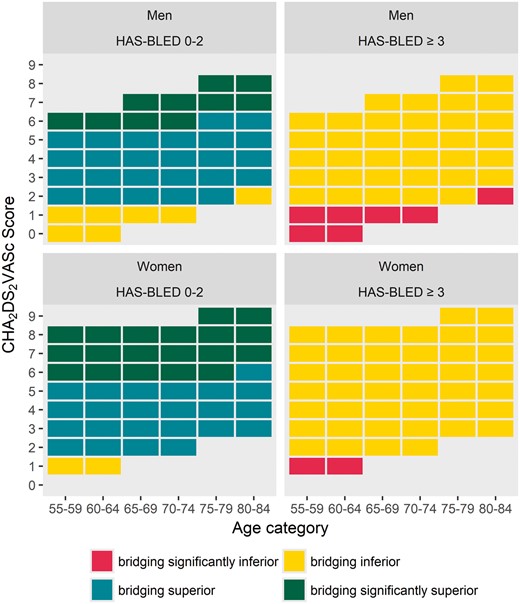
Bridging benefit decision matrix. Stratified by CHA2DS2-VASc and HAS-BLED scores, for various age categories and both sexes. CHA2DS2-VASc, congestive heart failure, hypertension, age, diabetes, stroke, transient ischaemic attack or thromboembolism, vascular disease, age and sex; HAS-BLED, hypertension, abnormal renal and liver function, stroke, bleeding, labile INR, elderly, drugs/alcohol.
For the base case (women 75–80 years old), Figure 4 displays the effect of the amount of days it takes to reach therapeutic INR values and the three different VKAs (warfarin, acenocoumarol, and phenprocoumon). Small differences were found between the three VKAs: at low risks of bleeding, bridging likely to be beneficial for patients on phenprocoumon from a CHA2DS2-VASc score of 5 compared with a score of 7 for patients on acenocoumarol. The benefit of bridging gets more pronounced when it takes longer to reach an INR of 2.5. If an INR of 2.5 was reached within 5 days, periprocedural bridging was never significantly beneficial, for both low and high bleeding risk patients. Reaching a therapeutic INR within 10 or 15 days marked the difference between having a significant benefit of periprocedural bridging at a CHA2DS2-VASc score of 5 or 4, respectively. The CHADS2 equivalents of Figure 4 are displayed in Supplementary material online, Figure S6.
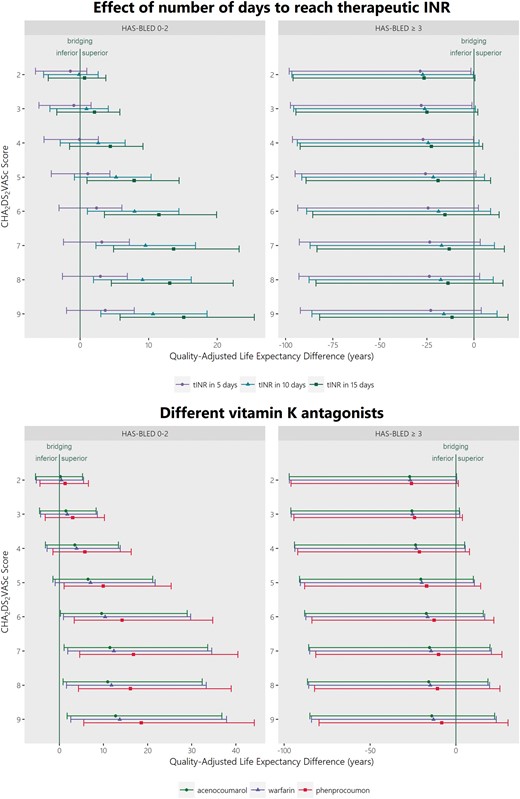
Effect of various vitamin K antagonists and time to reach therapeutic INR on quality-adjusted life expectancy difference of bridging. Stratified by CHA2DS2-VASc and HAS-BLED scores, results are for the base case, women of 75–80 years old, including the 95% confidence interval of the probabilistic sensitivity analysis. CHA2DS2-VASc, congestive heart failure, hypertension, age, diabetes, stroke, transient ischaemic attack or thromboembolism, vascular disease, age and sex; HAS-BLED, hypertension, abnormal renal and liver function, stroke, bleeding, labile INR, elderly, drugs/alcohol.
Discussion
The results of the base case analysis showed that stroke risk, bleeding risk, type of VKA, and time to reach therapeutic INR are important factors to consider, while deciding whether to apply periprocedural bridging anticoagulation. According to our evaluation, patients at a high risk of bleeding (HAS-BLED ≥3) are very unlikely to ever benefit from periprocedural bridging: the mean shows a decreased life expectancy in all cases, although usually not significant.
Patients with lower HAS-BLED scores may benefit if they have an elevated risk of stroke (CHA2DS2-VASc 6 or higher, CHADS2 4 or higher, 3 or higher for the age categories 55–65). Within the total AF population, around 18% of patients would have a sufficiently high stroke risk as defined by our calculated threshold value.10 Since the HAS-BLED score is not reported per CHA2DS2-VASc group, we do not know which proportion of this group would have a low HAS-BLED. The bleeding risk and stroke risk scores have corresponding predictors and consequently it is expected that only a very small number of patients with a high stroke risk would have a low bleeding risk. Therefore, we speculate that the patient group that could benefit from bridging anticoagulation according to our calculations, will be very small.
We found only slight differences between acenocoumarol, phenprocoumon, and warfarin. For patients with a low bleeding risk, bridging acenocoumarol is significantly beneficial from a CHA2DS2-VASc of 7 and higher, as opposed to a CHA2DS2-VASc of 6 and higher for warfarin and phenprocoumon. Our calculations stress the importance of post-procedural INR management: if patients reach a therapeutic INR within 5 days, strokes will occur less frequently, thus reducing the potential benefit of bridging. For patients in which it takes 10 or 15 days to reach an INR of 2.5, periprocedural bridging is only likely to be beneficial at higher CHA2DS2-VASc or CHADS2 scores. We expect the time to reach therapeutic INR will mainly depend on the patient-specific INR management, but it might also depend on the used VKA: e.g. for patients on phenprocoumon it may take longer to reach therapeutic INR.16 In clinical settings, the VKA used and the individual patient’s history regarding INR management could be taken into account when deciding whether to bridge or not.
Our results show a lot of uncertainty around the calculated means, especially for patients with high HAS-BLED scores. This is a result of the limited number of events, especially strokes, found within clinical studies. More real-life data could enhance the reliability of the results, for example, within the context of a large multicentre registry. The stroke risks in the model are calculated using the risk stratification schemes from the clinical setting to determine the necessity of anticoagulation, which may not be valid to use as a decision tool in surgical settings. Regarding the post-procedural stroke risk for AF patients, it would be preferable to use specific stratified stroke rates from the surgical setting, however, these numbers are not available.
The included strokes in the model are ischaemic, since most perioperative strokes are ischaemic instead of haemorrhagic, and data reliably differentiating ischaemic and haemorrhagic strokes is rare.17 Transient ischaemic attacks were not included in the model, because their the relative risk with warfarin treatment vs. non-treatment is not significant.18 Systemic embolisms were also not included, as the odds ratio of warfarin vs. placebo is not significant.19
The evidence for post-operative bleeding rates that incorporates both the HAS-BLED score and the effect of LMWHs is not available. This obstacle was tackled by using the effect of periprocedural bridging from the BRIDGE trial and the effect of the HAS-BLED score from Omran et al.3,11 Procedure-specific bleeding rates were not incorporated in the model, as the necessary data that could support this analysis, was not available in literature. The patient-specific bleeding rate, which we have included using HAS-BLED scores, can be used to approximate the procedure-specific bleeding rates: for procedures with high bleeding risks, bridging will be highly unlikely to be beneficial, while we may underestimate the benefit of bridging for low-risk procedures. However, for procedures with low bleeding risks, interrupting VKA treatment is not indicated, making our model superfluous.2 Thrombotic risk was not included in the model, since this is equal in both treatment arms.
The BRIDGE trial previously concluded that forgoing anticoagulation bridging is not inferior to perioperative bridging with LMWH for the prevention of arterial thromboembolism and decreased the risk of major bleeding.3 This evaluation demonstrated that for specific AF patients, bridging is expected to be beneficial. Within the BRIDGE trial, patients with relatively low stroke risks were included: CHADS2 2.3 (±1.03) and 2.4 (±1.07) for the non-bridging and bridging groups, respectively.3 These patients also do not benefit from periprocedural bridging in the base case of our simulation.
Dunn et al.4,6 previously found that bridging anticoagulation was preferred at an annual stroke rate of >5.6%, which would correspond to a CHADS2 score between 2 and 3. This outcome is comparable to our results, though in our model the difference is only significant from a CHADS2 score of 2 (age 55–65) or 4 (age 65–85). Compared with the article by Dunn et al.,3,6 we were able to incorporate more recent evidence to support the model, such as the BRIDGE trial. A more recent simulation study by Pappas et al.7 simulated net clinical benefit using population parameters for stroke and bleeding. As we used the quality adjusted life expectancy as the main outcome, we were able to take the long-term effects of strokes into account. Another difference is that we have incorporated increased risks, when compared with the population parameters, for bleeding and stroke post-procedurally.3,11
Current guidelines already advice to consider the risk of stroke, the patient-related bleeding risk and the bleeding risk of the procedure.2 The results of our model confirm this and, additionally, make it possible to identify more specific patient groups where bridging may be beneficial.
Our analysis stresses the importance of the post-procedural time to therapeutic INR. Limited research is available that focusses on the time it takes for AF patients to reach therapeutic INR levels after interrupting a VKA in the clinical setting. Frequent monitoring of the INR and tailored post-procedural VKA usage schemes seems to have a critical role in minimizing the risk of stroke. Currently, it is recommended that VKAs are reinitiated at the previous dose, however, there may be an opportunity to develop individualized dosing regimens to improve the time to reach therapeutic INR. Specifically, in the clinical setting, focusing on the optimal organization of post-procedural INR management for all VKA users may yield greater benefits than bridging the small subpopulation of VKA users that we identified may benefit from this.
Conclusion
In conclusion, our results show that only a small subset of AF patients is expected to benefit from bridging anticoagulation: those at a high risk of stroke (CHA2DS2-VASc ≥6, CHADS2 ≥4) and also at a low risk of bleeding (HAS-BLED ≤2).
Acknowledgements
The authors would like to thank the Regional Coagulation Roundtable of the Provinces Groningen and Drenthe (The Netherlands) and the Martini Hospital Coagulation Committee for their input during the development of the model.
Conflict of interest: R.G.T. reports grants and personal fees from Boehringer Ingelheim, personal fees from Bayer, personal fees from Bristol-Myers-Squibb, personal fees from Pfizer, outside the submitted work; M.J.P. received significant grants and modest honoraria from Boehringer Ingelheim, Bayer and BMS; has a modest share in Ingress Health and acts as an external advisor to ASC Academics, Groningen, the Netherlands and Mihajlovic Health Analytics, Novi Sad, Serbia. All other authors declared no conflict of interest. M.v.H. reports grants and personal fees from Boehringer Ingelheim and Bayer, outside the submitted work.
References
R Core Team.



