-
PDF
- Split View
-
Views
-
Cite
Cite
O Itzhaki Ben Zadok, D Leshem-Lev, T Ben-Gal, A Hamdan, N Schamroth-Pravda, T Steinmetz, I Kandinov, R Kornowski, A Eisen, Quantitative and functional evaluation of endothelial progenitor cells in patients with cardiac amyloidosis, European Heart Journal, Volume 41, Issue Supplement_2, November 2020, ehaa946.2114, https://doi.org/10.1093/ehjci/ehaa946.2114
Close - Share Icon Share
Abstract
Endothelial microvascular dysfunction is a known mechanism of injury in cardiac amyloidosis (CA), but evidence regarding the level and function of endothelial progenitor cells (EPCs) in patients with CA is lacking.
Study population included patients with light-chain or transthyretin (ATTR) CA. Patients with diagnosed heart failure and preserved ejection fraction (HFpEF) without monoclonal gammopathy and a 99mTc-DPD scan incompatible with TTR were used as controls. Blood circulating EPCs were assessed quantitatively by the expression of VEGFR-2(+), CD34(+) and CD133(+) using flow cytometry, and functionally by the formation of colony forming units (CFUs). MTT assay was used to demonstrate cell viability. Tests were repeated 3 months following the initiation of amyloid-suppressive therapies (either ATTR-stabilizer or targeted chemotherapy) in CA patients.
Our preliminary cohort included 14 CA patients (median age 74 years, 62% ATTR CA).
Patients with CA vs. patients with HFpEF (n=8) demonstrated lower expression of CD34(+)/VEGFR-2(+) cells [0.51% (IQR 0.4, 0.7) vs. 1.03% (IQR 0.6, 1.4), P=0.043] and CD133(+)/VEGFR-2(+) cells [0.35% (IQR 0.23, 0.52) to 1.07% (IQR 0.6, 1.5), P=0.003]. Functionally, no differences were noted between groups. Following the initiation of amyloid-suppressive therapies in CA patients, we observed the up-regulation of CD34(+)/VEGFR-2(+) cells [2.47% (IQR 2.1, 2.7), P<0.001] and CD133(+)/VEGFR-2(+) cells [1.38% (IQR 1.1, 1.7), P=0.003]. Moreover, functionally, active EPCs were evident microscopically by their ability to form colonies (from 0.5 CFUs [IQR 0, 1.5) to 2 CFUs (IQR 1, 3.5), P=0.023]. EPCs' viability was demonstrated by an MTT assay [0.12 (IQR 0.04, 0.12) to 0.24 (IQR 0.16, 0.3), p=0.014].
These preliminary results demonstrate reduced EPCs levels in CA patients indicating significant microvascular impairment. Amyloid-targeted therapies induce the activation of EPCs, thus possibly promoting endothelial regeneration. These findings may represent a novel mechanism of action of amyloid-suppressive therapies
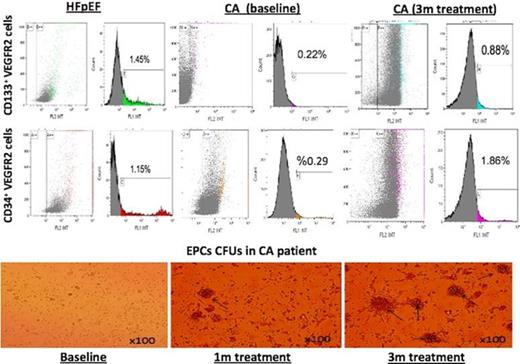
EPCs in CA patients and during therapy
Type of funding source: None
- amyloid
- technetium 99m
- cardiomyopathy
- endothelial progenitor cells
- vascular endothelial growth factor receptor-2
- chemotherapy regimen
- cardiac amyloidosis
- flow cytometry
- cd34 antigens
- antigens, cd98 light chains
- cell survival
- endothelium
- paraproteinemias
- up-regulation (physiology)
- pharmacokinetics
- prealbumin
- monoclonal gammopathy of undetermined significance
- heart failure with preserved ejection fraction
- mechanism of injury
- stabilizer ingredient



