-
PDF
- Split View
-
Views
-
Cite
Cite
Seyed Reza Saghebi, Mahdi Zangi, Tissa Tajali, Roya Farzanegan, Seyed Mehdi Farsad, Azizollah Abbasidezfouli, Kambiz Sheikhy, Mohammad Behgam Shadmehr, The role of T-tubes in the management of airway stenosis, European Journal of Cardio-Thoracic Surgery, Volume 43, Issue 5, May 2013, Pages 934–939, https://doi.org/10.1093/ejcts/ezs514
Close - Share Icon Share
Abstract
When the T-tube is inserted as a temporary stent, it is unclear whether keeping it longer in place has any benefit on the outcome.
Among 1738 patients with airway stenosis (1996–2011), 134 underwent T-tube placement (mean duration = 14.3 months); temporarily while waiting for an appropriate time for surgery in 53 (Group 1), as an adjunct after a complex laryngotracheal resection in 27 (Group 2), after surgical failure in 43 (Group 3) and permanently in 11 unresectable strictures (Group 4). A logistic regression model was used for statistical analysis.
Seventy percent of patients were males (age = 33.6 ± 17 years). The main cause was postintubation/post-tracheostomy stenosis in 87% of patients. The stenosis (29.6 ± 14 mm, 5–80 mm) was located in the subglottis in 33%, trachea in 47% and both in 20% of cases. To assess the effect of T-tubes on stabilizing the airway after decannulation, 50 patients who still had a T-tube at the end of follow-up or for <1.5 months were excluded. Of the remaining 84, 31.5, 91.5 and 32.5% of patients in Groups 1, 2 and 3 were stable at least 3 months after decannulation. Moreover, 70% of those who were decannulated at or before 6 months and 53.7% of those who were decannulated after 6 months underwent another intervention (P = 0.17). The age, sex, cause, site of stenosis and even duration of T-tube insertion (P = 0.07) showed no significant effect on the decannulation outcome.
Although it seems that keeping the T-tube in place for >6 months may increase the chance of successful decannulation, it was not confirmed in our study.
INTRODUCTION
The most common cause of tracheal stenosis is prolonged intubation [1, 2]. The other, but infrequent, causes are direct airway trauma, tumours, inhalation injuries, infections (like tuberculosis), idiopathic stenosis and congenital malformations [2]. The best treatment for tracheal stenosis is surgical resection and reconstruction, if feasible [3]. However, it is not possible forever in some cases because of the long length of the destructed segment of trachea, or severe co-morbidities [4]. In some other patients, it is not logical to do the surgical resection at the time of diagnosis, and it is better to postpone it for a more appropriate time [5]. The examples are trauma victims with neurological sequelae that require more time to become fixed or at least stable, those with associated orthopaedic or maxillofacial injuries who are supposed to undergo surgical procedures under general anaesthesia, those who had tracheal stenosis because of prolonged intubation for myocardial infarction or cerebrovascular accidents and need more time for recovery and also some patients with multisegmental tracheal stenosis [6].
In such circumstances, and sometimes after complex airway surgical procedures, we have to temporarily or permanently stent the airway [5, 7]. The simplest and most practical stent is tracheostomy, but it is not the best. Tracheostomy not only impairs normal phonation, but also diverts the passage of air through the nose and larynx directly to the trachea and, therefore, exposes the major airways and lungs to dried air, leading to chronic cough and secretions. It is also irritating to the mucosa [8]. To overcome these disadvantages, Dr Montgomery [9] from Harvard Medical School introduced silicone T-tubes in 1965. Since then, several articles have been published regarding the indications, advantages, complications and outcome of managing tracheal stenosis with T-tubes.
When the T-tube is used as a temporary stent, it was shown that some patients would not need further surgical airway resection after decannulation [2, 5, 8]. It seems that the injured segment of trachea accomplishes its maturation over time and makes a cicatricial tubular structure all around the T-tube, and when the T-tube is removed, the trachea remains patent and stable. However, it has not yet been made clear, for how long the T-tube should be kept in place, and whether keeping it longer in the trachea has any benefit on outcomes or not.
We tried to answer this question by a retrospective analysis of our patients who were managed with T-tubes.
PATIENTS AND METHODS
Patients
We reviewed our General Thoracic Surgery Ward Database and found that 134 (7.7%) patients, among 1738 patients, with non-tumoural tracheal stenosis were managed with a T-tube during a 15-year period from April 1996 to October 2011. The Medical Ethics Committee approved the study (Medical Ethics code number: 055).
Treatment groups
T-tubes were inserted in four groups of patients.
Group 1 consisted of 53 patients who were not suitable candidates for airway resection at the time of diagnosis. There were some reasons, including the following factors:
Patients with neurological sequelae, myocardial Infarction, cerebrovascular accidents or severe pulmonary insufficiency who required more time to become stable and cooperative to undergo airway surgery.
Patients with associated orthopaedic or maxillofacial injuries who were supposed to undergo another surgical procedure under general anaesthesia, and we did not want to expose a fresh airway anastomosis to intubation.
Patients with very long-length (>50%) tracheal stenosis or malacia, multisegmental tracheal stenosis or severe airway inflammation and infection.
The patients with severe glottic/subglottic injuries who required more time for subsidence of inflammation and recovery of neuromuscular function of their larynx, to make a final decision regarding the patency of the upper airway before any tracheal resection.
The T-tube was temporarily inserted in this group of patients as a bridge, while waiting for an appropriate time for surgical resection.
Group 2 consisted of 27 patients who underwent a complex laryngotracheal resection and reconstruction, and we were concerned about the postoperative airway patency. The examples were laryngotracheal resection with complex reconstruction at the level of glottis and subglottis, laryngotracheal resection and reconstruction in children, and resection and reconstruction of one stricture in multisegmental tracheal stenosis.
T-tube was temporarily inserted as an adjunct in this group of patients.
Group 3 consisted of 43 patients with recurrence after an airway resection and reconstruction. As our policy is to perform airway re-resection about 6 months after the first attempt, T-tubes were temporarily inserted in this group of patients, to keep them away from repeated bronchoscopic dilatations.
Group 4 consisted of 11 patients in whom T-tube was inserted as a permanent stent because of either a very lengthy stricture or severe and unstable co-morbid diseases.
T-tube insertion procedure
All patients underwent thorough laryngeal evaluation by local anaesthesia, intravenous sedation and then tracheal evaluation by general anaesthesia, before T-tube insertion in the operating room.
The anatomy and function of vocal cords, the length of the stenotic segment, the distance of its proximal end to the vocal cords and its distal end to carina, as well as the size and location of stoma (if present) in relation to the stenotic segment were all precisely determined. The diameter of the T-tube was chosen to be similar to the diameter of the largest rigid scope that had dilated the stenotic segment. Then its proximal and distal vertical branches were trimmed based on the length of stenosis and its distance to the vocal cords and carina.
The procedures were performed by two thoracic surgeons; one sitting above the table and the other inserting the tube through the stoma. When there was a tracheostomy stoma, in the vast majority of cases, we had to dilate the stoma by Hegar bougies or by a small incision. Otherwise, the procedure was performed via a small transverse collar incision like a tracheostomy incision.
Before tube insertion, ventilation was maintained through the rigid scope, and during tube insertion it was continued through a number 4 or 5 cuffless endotracheal tube that had been passed through the horizontal branch to the distal vertical branch (Fig. 1).
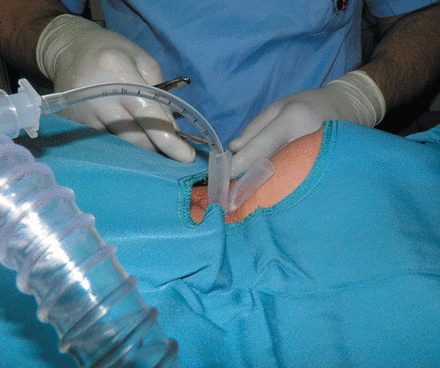
After tube placement, its proper position was confirmed by 0° telescope or fibreoptic bronchoscope, and the horizontal branch was capped as soon as the patient was able to breathe spontaneously and expectorate secretions through the mouth. In some patients, irrespective of precise bronchoscopic measurements, we had to remove the tube once or a couple of times and cut more and more either proximally or distally for a better adjustment.
In patients in whom the proximal vertical branch of the T-tube was passed through and up to their vocal cords, the proximal aperture was sometimes capped temporarily to prevent early postoperative aspiration, and obviously the horizontal branch was kept open. After several days, the cap was removed and the majority of these patients were able to eat and drink without any significant aspiration.
During the first week after T-tube insertion, most patients underwent frequent toilet bronchoscopy by flexible scopes. Before discharge, all patients and their family members or home nurses were taught how to suction the T-tube, whenever it might be required and more importantly, how to remove the T-tube in case of emergent occlusion and replace it with a tracheostomy tube. All patients were asked to actively participate in their scheduled follow-up programme including office visits and/or bronchoscopy based on their underlying disease.
Data analysis
SPSS for windows (version 16.0) software was used to input and analyse the data of this study. Chi-square (χ2), Fisher's exact test, independent sample t-test and multivariate logistic regression were applied to analyse the data. P-values <0.05 were considered as the significant levels.
Follow-up
Follow-ups were completed by chart review, direct patients' or their family contacts and office visits. After T-tube removal, the average duration of follow-up was 8.8 months.
RESULTS
Demographic data
There were 94 (70%) males and 40 (30%) females, with a mean age of 33.6 ± 17 years and a range of 6–81 years.
Stenosis characteristics
The different causes of airways stenosis in our patients are shown in Table 1. Postintubation/post-tracheostomy stenosis was the most common aetiology. The mean duration of orotracheal intubation in this group of patients was 15.2 (range 2–30) days.
| Cause of stenosis . | Number . | Percent . |
|---|---|---|
| Prolonged intubation | 116 | 86.6 |
| Airway trauma | 7 | 5.2 |
| Idiopathic | 3 | 2.2 |
| Othersa | 8 | 6 |
| Cause of stenosis . | Number . | Percent . |
|---|---|---|
| Prolonged intubation | 116 | 86.6 |
| Airway trauma | 7 | 5.2 |
| Idiopathic | 3 | 2.2 |
| Othersa | 8 | 6 |
aTuberculosis, rheumatoid arthritis, inhalation injuries, missing.
| Cause of stenosis . | Number . | Percent . |
|---|---|---|
| Prolonged intubation | 116 | 86.6 |
| Airway trauma | 7 | 5.2 |
| Idiopathic | 3 | 2.2 |
| Othersa | 8 | 6 |
| Cause of stenosis . | Number . | Percent . |
|---|---|---|
| Prolonged intubation | 116 | 86.6 |
| Airway trauma | 7 | 5.2 |
| Idiopathic | 3 | 2.2 |
| Othersa | 8 | 6 |
aTuberculosis, rheumatoid arthritis, inhalation injuries, missing.
The mean length of stenosis was 29 ± 14.0 mm with a range of 5–80 mm. The stenosis was located only in the subglottic trachea in 33%, in the trachea in 47% and in both laryngotracheal segments in 20% of cases.
The main gross pathological feature of the stenotic segment was fibrotic stricture in the vast majority of cases (80.2%). However, it was associated with malacia and/or granulation tissue in 35.2% of them. The remainder was either just granulation tissue or malacia in nature. Approximately 9% of all patients had multisegmental tracheal stenosis.
Treatment groups
Table 2 summarizes some important data and outcome of our patients in each individual group. There were 43 (32.1%) complications (Table 3); 16 resulted in T-tube failure and its replacement with the tracheostomy tube.
| Type . | No. . | T-tube duration (month) . | Tolerated decannulation . | Resection and reconstruction after decannulation . | Still having T-tube on follow-up . | Tracheostomy . | Mortality . |
|---|---|---|---|---|---|---|---|
| Group 1 | 53 | 9.6 | 14 (26.4%) | 23 (43.4%) | 5 (9.4%) | 8 (15.2%) | 2 (3.8%) |
| Group 2 | 27 | 8.9 | 15 (55.5%) | 1 (3.7%) | 6 (22.2%) | 3 (11%) | 1 (3.7%) |
| Group 3b | 43 | 20 | 13 (30.2%) | 20 (46.5%) | 6 (14%) | 3 (7%) | 0 |
| Group 4 | 11 | 28.8 | 1 (9%) | 0 | 6 (54.5%) | 2 (18%) | 1 (9%) |
| Type . | No. . | T-tube duration (month) . | Tolerated decannulation . | Resection and reconstruction after decannulation . | Still having T-tube on follow-up . | Tracheostomy . | Mortality . |
|---|---|---|---|---|---|---|---|
| Group 1 | 53 | 9.6 | 14 (26.4%) | 23 (43.4%) | 5 (9.4%) | 8 (15.2%) | 2 (3.8%) |
| Group 2 | 27 | 8.9 | 15 (55.5%) | 1 (3.7%) | 6 (22.2%) | 3 (11%) | 1 (3.7%) |
| Group 3b | 43 | 20 | 13 (30.2%) | 20 (46.5%) | 6 (14%) | 3 (7%) | 0 |
| Group 4 | 11 | 28.8 | 1 (9%) | 0 | 6 (54.5%) | 2 (18%) | 1 (9%) |
aThree patients missed the follow-up.
bOne patient in Group 3 was treated by repeated laser.
| Type . | No. . | T-tube duration (month) . | Tolerated decannulation . | Resection and reconstruction after decannulation . | Still having T-tube on follow-up . | Tracheostomy . | Mortality . |
|---|---|---|---|---|---|---|---|
| Group 1 | 53 | 9.6 | 14 (26.4%) | 23 (43.4%) | 5 (9.4%) | 8 (15.2%) | 2 (3.8%) |
| Group 2 | 27 | 8.9 | 15 (55.5%) | 1 (3.7%) | 6 (22.2%) | 3 (11%) | 1 (3.7%) |
| Group 3b | 43 | 20 | 13 (30.2%) | 20 (46.5%) | 6 (14%) | 3 (7%) | 0 |
| Group 4 | 11 | 28.8 | 1 (9%) | 0 | 6 (54.5%) | 2 (18%) | 1 (9%) |
| Type . | No. . | T-tube duration (month) . | Tolerated decannulation . | Resection and reconstruction after decannulation . | Still having T-tube on follow-up . | Tracheostomy . | Mortality . |
|---|---|---|---|---|---|---|---|
| Group 1 | 53 | 9.6 | 14 (26.4%) | 23 (43.4%) | 5 (9.4%) | 8 (15.2%) | 2 (3.8%) |
| Group 2 | 27 | 8.9 | 15 (55.5%) | 1 (3.7%) | 6 (22.2%) | 3 (11%) | 1 (3.7%) |
| Group 3b | 43 | 20 | 13 (30.2%) | 20 (46.5%) | 6 (14%) | 3 (7%) | 0 |
| Group 4 | 11 | 28.8 | 1 (9%) | 0 | 6 (54.5%) | 2 (18%) | 1 (9%) |
aThree patients missed the follow-up.
bOne patient in Group 3 was treated by repeated laser.
| . | Frequency . | Percent . |
|---|---|---|
| T-tube obstruction due to mucus plug formation | 18 | 13.4 |
| Proximal granulation tissue | 16 | 11.9 |
| Aspiration | 6 | 4.5 |
| Distal granulation tissue | 2 | 1.5 |
| Displacement of T-tube | 1 | 0.7 |
| Total | 43 | 32.1 |
| . | Frequency . | Percent . |
|---|---|---|
| T-tube obstruction due to mucus plug formation | 18 | 13.4 |
| Proximal granulation tissue | 16 | 11.9 |
| Aspiration | 6 | 4.5 |
| Distal granulation tissue | 2 | 1.5 |
| Displacement of T-tube | 1 | 0.7 |
| Total | 43 | 32.1 |
| . | Frequency . | Percent . |
|---|---|---|
| T-tube obstruction due to mucus plug formation | 18 | 13.4 |
| Proximal granulation tissue | 16 | 11.9 |
| Aspiration | 6 | 4.5 |
| Distal granulation tissue | 2 | 1.5 |
| Displacement of T-tube | 1 | 0.7 |
| Total | 43 | 32.1 |
| . | Frequency . | Percent . |
|---|---|---|
| T-tube obstruction due to mucus plug formation | 18 | 13.4 |
| Proximal granulation tissue | 16 | 11.9 |
| Aspiration | 6 | 4.5 |
| Distal granulation tissue | 2 | 1.5 |
| Displacement of T-tube | 1 | 0.7 |
| Total | 43 | 32.1 |
Group 1
There were 53 patients in this group. The mean duration of T-tube placement was 9.6 months. T-tubes were removed in all patients in the operating room, and the laryngeal anatomy and function as well as the tracheal anatomy were completely re-evaluated.
In 14 (26.4%) patients, a patent and stable airway was observed. They were kept decannulated and followed for at least 3 months, and all were stable at the end of follow-up. However, 23 (43.4%) patients underwent airway resection and anastomosis after decannulation.
In 5 (9.4%) patients, T-tubes were re-inserted because the main goal of using the T-tube as a bridge had not yet been achieved.
Two (3.8%) patients died in this group; one because of myocardial infarction and another due to an unknown reason.
Group 2
There were 27 patients in this group. The mean duration of T-tube placement was 8.9 months. Fifteen (55.5%) patients tolerated decannulation well, while 6 (22.2%) did not cope with it and underwent T-tube re-insertion. Only 1 (3.7%) patient underwent airway re-resection and anastomosis after decannulation. Unfortunately, 1 (3.7%) patient died because of T-tube occlusion by mucus plug.
Group 3
There were 43 patients in this group with a mean duration of T-tube placement of 20 months. In 13 (30.2%) patients, a patent and stable airway was observed after decannulation and no subsequent surgical intervention was required. However, in 20 (46.5%) patients, airway re-resection and anastomosis were performed because of enduring stenosis.
In 6 (14%) patients, T-tubes were re-inserted because of nearly no chance of re-resection at that time and hope for the improvement of condition in the future.
Group 4
There were 11 patients in this group. The mean duration of T-tube placement was 28.8 months. One (9%) patient tolerated decannulation well, while 6 (54.5%) did not cope with it and underwent T-tube re-insertion.
One (9%) patient died due to the progression of her advanced cardiac failure.
Risk factors and T-tube decannulation
To assess the effect of T-tube on stabilizing the airway and to eliminate the necessity for further intervention after decannulation, 50 patients who still had T-tube at the end of follow-up or had T-tubes for <1.5 months were excluded. Of the remaining 84, 31.5, 91.5 and 32.5% of patients in Groups 1, 2 and 3 were stable at least 3 months after decannulation with no further intervention (P-value <0.001; Fig. 2).
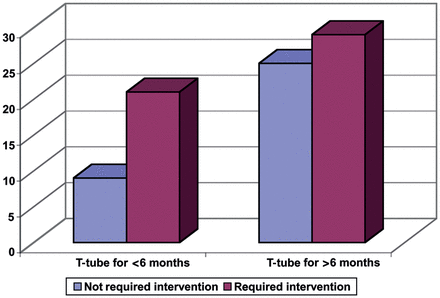
Need for further intervention based on the duration of T-tube.
Bivariate analysis of all potentially contributing factors (except the length and diameter of the stenosis that had about one-third and one-half missing data, respectively) in determining the need for further intervention after decannulation is shown in Table 4. To make sure that the finding of the bivariate analysis is reliable, all data were entered in a logistic regression model (Table 5). The whole logistic model was significant (Hosmer and Lemeshow test: significance = 0.52).
Bivariate analysis of the possible predictive variables of the decannulation outcome
| Factors . | Further intervention after decannulation (%) . | P-value . |
|---|---|---|
| Age | NS | |
| Sex | ||
| Female | 70 | NS |
| Male | 56.2 | |
| Cause of stenosis | ||
| Postintubation/post-tracheostomy | 58.9 | NS |
| Other causes | 63.6 | |
| Site of stenosis | ||
| Subglottic and laryngotracheal stenosis | 51.9 | NS |
| Tracheal stenosis | 63.8 | |
| Duration of T-tube | ||
| T-tube insertion ≤6 months | 70 | NS |
| T-tube insertion >6 months | 53.7 | |
| Treatment group | ||
| Group 1 | 68.4 | <0.001 |
| Group 2 | 8.3 | |
| Group 3 | 67.6 | |
| Factors . | Further intervention after decannulation (%) . | P-value . |
|---|---|---|
| Age | NS | |
| Sex | ||
| Female | 70 | NS |
| Male | 56.2 | |
| Cause of stenosis | ||
| Postintubation/post-tracheostomy | 58.9 | NS |
| Other causes | 63.6 | |
| Site of stenosis | ||
| Subglottic and laryngotracheal stenosis | 51.9 | NS |
| Tracheal stenosis | 63.8 | |
| Duration of T-tube | ||
| T-tube insertion ≤6 months | 70 | NS |
| T-tube insertion >6 months | 53.7 | |
| Treatment group | ||
| Group 1 | 68.4 | <0.001 |
| Group 2 | 8.3 | |
| Group 3 | 67.6 | |
NS: not significant.
Bivariate analysis of the possible predictive variables of the decannulation outcome
| Factors . | Further intervention after decannulation (%) . | P-value . |
|---|---|---|
| Age | NS | |
| Sex | ||
| Female | 70 | NS |
| Male | 56.2 | |
| Cause of stenosis | ||
| Postintubation/post-tracheostomy | 58.9 | NS |
| Other causes | 63.6 | |
| Site of stenosis | ||
| Subglottic and laryngotracheal stenosis | 51.9 | NS |
| Tracheal stenosis | 63.8 | |
| Duration of T-tube | ||
| T-tube insertion ≤6 months | 70 | NS |
| T-tube insertion >6 months | 53.7 | |
| Treatment group | ||
| Group 1 | 68.4 | <0.001 |
| Group 2 | 8.3 | |
| Group 3 | 67.6 | |
| Factors . | Further intervention after decannulation (%) . | P-value . |
|---|---|---|
| Age | NS | |
| Sex | ||
| Female | 70 | NS |
| Male | 56.2 | |
| Cause of stenosis | ||
| Postintubation/post-tracheostomy | 58.9 | NS |
| Other causes | 63.6 | |
| Site of stenosis | ||
| Subglottic and laryngotracheal stenosis | 51.9 | NS |
| Tracheal stenosis | 63.8 | |
| Duration of T-tube | ||
| T-tube insertion ≤6 months | 70 | NS |
| T-tube insertion >6 months | 53.7 | |
| Treatment group | ||
| Group 1 | 68.4 | <0.001 |
| Group 2 | 8.3 | |
| Group 3 | 67.6 | |
NS: not significant.
Regression model of the possible predictive variables of the decannulation outcome
| Factors . | P-value . | Odds ratio . | 95% CI . | |
|---|---|---|---|---|
| Lower . | Upper . | |||
| Age* | 0.312 | 0.981 | 0.944 | 1.019 |
| Sex | ||||
| Male | 0.359 | 0.521 | 0.129 | 2.1 |
| Female | 1 | |||
| Cause of stenosis | ||||
| Other causes | 0.523 | 1.811 | 0.293 | 11.191 |
| Postintubation/post-tracheostomy | 1 | |||
| Site of stenosis | ||||
| Subglottic and laryngotracheal stenosis | 0.306 | 0.559 | 0.184 | 1.7 |
| Tracheal stenosis | 1 | |||
| Duration of T-tube | ||||
| T-tube insertion >6 months | 0.07 | 0.271 | 0.066 | 1.112 |
| T-tube insertion ≤6 months | 1 | |||
| Treatment group | ||||
| Group 1 | 0.457 | 0.627 | 0.183 | 2.147 |
| Group 2 | 0.004 | 0.029 | 0.002 | 0.331 |
| Group 3 | 1 | |||
| Factors . | P-value . | Odds ratio . | 95% CI . | |
|---|---|---|---|---|
| Lower . | Upper . | |||
| Age* | 0.312 | 0.981 | 0.944 | 1.019 |
| Sex | ||||
| Male | 0.359 | 0.521 | 0.129 | 2.1 |
| Female | 1 | |||
| Cause of stenosis | ||||
| Other causes | 0.523 | 1.811 | 0.293 | 11.191 |
| Postintubation/post-tracheostomy | 1 | |||
| Site of stenosis | ||||
| Subglottic and laryngotracheal stenosis | 0.306 | 0.559 | 0.184 | 1.7 |
| Tracheal stenosis | 1 | |||
| Duration of T-tube | ||||
| T-tube insertion >6 months | 0.07 | 0.271 | 0.066 | 1.112 |
| T-tube insertion ≤6 months | 1 | |||
| Treatment group | ||||
| Group 1 | 0.457 | 0.627 | 0.183 | 2.147 |
| Group 2 | 0.004 | 0.029 | 0.002 | 0.331 |
| Group 3 | 1 | |||
*Age was considered as a continuous variable.
Regression model of the possible predictive variables of the decannulation outcome
| Factors . | P-value . | Odds ratio . | 95% CI . | |
|---|---|---|---|---|
| Lower . | Upper . | |||
| Age* | 0.312 | 0.981 | 0.944 | 1.019 |
| Sex | ||||
| Male | 0.359 | 0.521 | 0.129 | 2.1 |
| Female | 1 | |||
| Cause of stenosis | ||||
| Other causes | 0.523 | 1.811 | 0.293 | 11.191 |
| Postintubation/post-tracheostomy | 1 | |||
| Site of stenosis | ||||
| Subglottic and laryngotracheal stenosis | 0.306 | 0.559 | 0.184 | 1.7 |
| Tracheal stenosis | 1 | |||
| Duration of T-tube | ||||
| T-tube insertion >6 months | 0.07 | 0.271 | 0.066 | 1.112 |
| T-tube insertion ≤6 months | 1 | |||
| Treatment group | ||||
| Group 1 | 0.457 | 0.627 | 0.183 | 2.147 |
| Group 2 | 0.004 | 0.029 | 0.002 | 0.331 |
| Group 3 | 1 | |||
| Factors . | P-value . | Odds ratio . | 95% CI . | |
|---|---|---|---|---|
| Lower . | Upper . | |||
| Age* | 0.312 | 0.981 | 0.944 | 1.019 |
| Sex | ||||
| Male | 0.359 | 0.521 | 0.129 | 2.1 |
| Female | 1 | |||
| Cause of stenosis | ||||
| Other causes | 0.523 | 1.811 | 0.293 | 11.191 |
| Postintubation/post-tracheostomy | 1 | |||
| Site of stenosis | ||||
| Subglottic and laryngotracheal stenosis | 0.306 | 0.559 | 0.184 | 1.7 |
| Tracheal stenosis | 1 | |||
| Duration of T-tube | ||||
| T-tube insertion >6 months | 0.07 | 0.271 | 0.066 | 1.112 |
| T-tube insertion ≤6 months | 1 | |||
| Treatment group | ||||
| Group 1 | 0.457 | 0.627 | 0.183 | 2.147 |
| Group 2 | 0.004 | 0.029 | 0.002 | 0.331 |
| Group 3 | 1 | |||
*Age was considered as a continuous variable.
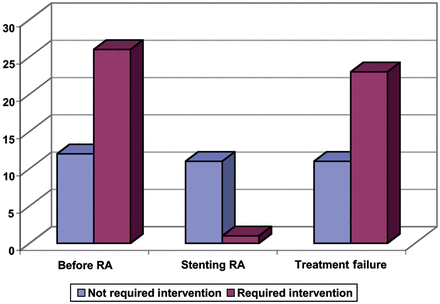
As expected, the regression model drew a significant distinction between Group 2 and other groups in terms of the decannulation outcome (P-value = 0.004). The P-value for the effect of ‘T-tube duration’ was calculated at 0.07 (Fig. 3).
Furthermore, variation of the decannulation outcome is predicted by the variables of this model by 30.6% (Nagelkerke R2: 0.306).
DISCUSSION
The best treatment for laryngotracheal stenosis is segmental airway resection and reconstruction [3]. However, it is not always possible at the time of diagnosis due to many different reasons related either to the airway itself or associated co-morbid diseases [4]. In such a situation, one alternative for the short-term preservation of airway patency is repeated bronchoscopic core-out of granulation tissues and dilatation of stricture under general anaesthesia, but if the surgical resection is required to be postponed for a notable amount of time, it would be impractical both for the patient and the surgeon. Therefore, we would need a tracheal stent, which could be left in the airway for a long time with minimum complications.
Such an ideal stent should be rigid enough to resist external pressure, but somehow soft and flexible to be minimally irritating to laryngotracheal mucosa. On the other hand, it should stay fixed in place in order not to be dislodged by coughing, although it ought to be easily removable when the time of surgery is due or in emergency situations [10, 11]. To catch these advantages as well as enjoy the normal phonation and humidification of the airway, we prefer to stent our patients with T-tubes rather than tracheostomy tubes, Dumon silicone stents, polyflex stents or expandable metallic stents [12–14]. Another unique advantage of T-tubes is that they are the only stents that could be used for the strictures at the level of the vocal cords and subglottic trachea. On the other hand, a disadvantage that was also noted in our patients was its occlusion by dried mucus and sputum [15]. Besides, when this happens at home and is not relieved by suction, T-tube removal is not as convenient as tracheostomy tube removal for the patients or their family members.
The principles of our method for T-tube insertion are more or less the same as others [5, 7, 8, 16], but some subtle details of our technique like keeping all patients under general anaesthesia with a muscle relaxant allowed us to do the procedure very gently with no intraoperative injury or asphyxia.
T-tubes were used in four different groups of patients. In Groups 1 and 3, it was initially used as a temporary therapeutic management in 53 and 43 of our patients, respectively, to buy some time and reach a more suitable time for a definitive surgical airway resection or re-resection in a variety of clinical situations. At the end of follow-up, our results demonstrated quite a successful outcome for these two groups of patients, with a low failure (change to tracheostomy tube) rate (15.2 and 7%, respectively) and minimal mortality rate (3.8 and 0%, respectively). Moreover, 23 (43.4%) patients in Group 1 and 20 (46.5%) in Group 3 underwent airway resection and reconstruction at an appropriate time. More importantly, 14 (26.4%) patients in Group 1 and 13 (30.2%) in Group 3 became decannulated with patent and stable airways, and no further interventions were required for them, therefore, the T-tube played the role of a definitive treatment for these patients. This great advantage of the T-tube has been previously shown by most studies [2, 5, 8, 17], although it was not consistent with one of our reviewed studies [4], which might be due to patients' conditions or selection. In these groups of patients, the reason for which the surgical resection has been delayed dictates the proper time for a definitive operation. For instance, whenever we have a patient who recently underwent a tracheal resection and reconstruction, and the anastomosis has developed a dehiscence, a rigid bronchoscopy is first performed to confirm the diagnosis, core-out the granulation tissues and dilate the anastomosis. If the symptoms and signs of airway stenosis recur soon and repeated bronchoscopic dilatation is required, we would rather insert a T-tube and wait for at least 6 months after the first attempt at surgical resection [18, 19]. After decannulation at the desired time, if the stenosis is still present and resectable, a second surgery will be definitively performed. But when its resectability is borderline, then the challenge is whether we have to wait longer or take the risk of re-resection at the moment and not waste time for the patient. Gaissert et al. [8] had a patient in their series who underwent successful stent extraction 9 years after the first surgery. We had some patients in the paediatric age group and gave them more time, because we believed that by their airway growth, there would be more chance for a successful decannulation. There are also several more examples such as when we decannulate a patient who had previously undergone T-tube placement for a lengthy tracheal stenosis due to fibrosis or infection and inflammation; and we notice a significant shortening of the stenotic segment, but there is still some remaining inflammation and infection and the resectability is borderline. Again, it is unclear if waiting longer has any benefit for the patient or not.
To answer these questions, thorough bivariate and then multivariate analyses of all potentially contributing factors for a successful decannulation were conducted. One-third and one-half of the data were missing in two of the eight possible predictive factors: the length of stenosis and the diameter of stenosis, respectively. This was because our hospital is a national referral centre for airway diseases, and the initial details of the stenosis characteristics of quite a considerable number of patients were unavailable. According to a well-known rule of thumb, or in other words, the 1 in 10 rule [20], there were limitations to entering all the predictive factors in the regression model to assess the effective ones in decannulation outcomes. Despite this fact, and also the possibility of an association between the longitudinal extent of stenosis and decannulation outcome shown in some other studies [21], we entered the length and diameter of stenosis in an alternative regression model besides the site of stenosis, T-tube duration and treatment groups. Similar to our findings in the main regression model, none of the factors except the ‘treatment group 2’ was associated with the decannulation outcome.
Our results illustrated that most patients in Group 2 tolerated decannulation quite well. This is not surprising, because they were patients who underwent a complex laryngotracheal resection and reconstruction, and our serious concerns were early postoperative upper airway oedema and late postoperative anastomotic stricture. Therefore, a T-tube was inserted intraoperatively, and fortunately, it resulted in excellent outcomes in our patients.
One interesting finding, even though statistically not significant (P = 0.07) was the role of ‘T-tube duration’ in decannulation outcomes, which was the main question of our study. Perhaps if we repeat our study after several years with more patients, a more reliable answer will be achieved.
Patients in Group 4 were not entered in our regression model, because they were non-homogenous to other groups. While 1 patient became and remained decannulated, they did not have any chance of undergoing airway resection and reconstruction in case of not tolerating decannulation.
There were 43 (32.1%) complications in our patients, most commonly T-tube obstruction by mucus plug formation in 18 patients and granulation tissue formation at the top of the proximal or below the distal vertical branches in 16 and 2 patients, respectively. All these patients were managed with bronchoscopy, although some of them resulted in T-tube failure due to frequent occurrence of the complication [7, 8, 10]. There were four mortalities, three of them unrelated to T-tube insertion, but unfortunately one (0.7%) suffocation happened due to T-tube obstruction with mucus plug formation.
Although it was not confirmed in our study that keeping the T-tube longer in place has any significant advantage on outcomes and there is also no consensus in the literature regarding this time duration [2, 22], it is recommended that when a patient does not tolerate T-tube removal at the scheduled time, it should not be considered as a definitive failure. If the ideal conditions for surgical resection of stenosis have been achieved, we highly recommend carrying out the operation because of its high success rate and low morbidity and mortality rates, otherwise the T-tube could be inserted and removed several more times with the hope of fixing the airway and keeping it patent before considering the patient as an obligatory life-long T-tube user.
Conflict of interest: none declared.
REFERENCES
Author notes
Presented at the 20th European Conference on General Thoracic Surgery, Essen, Germany, 10–13 June 2012.




