-
PDF
- Split View
-
Views
-
Cite
Cite
G. Riest, J. Peters, M. Weiss, S. Dreyer, P. D. Klassen, B. Stegen, A. Bello, M. Eikermann, Preventive effects of perioperative parecoxib on post-discectomy pain, BJA: British Journal of Anaesthesia, Volume 100, Issue 2, February 2008, Pages 256–262, https://doi.org/10.1093/bja/aem345
Close - Share Icon Share
Abstract
Cyclooxygenase inhibitor treatment is viewed increasingly critical because of safety considerations, and there are several open questions on their optimal use.
In a randomized placebo-controlled study in 320 patients undergoing discectomy, we administered parecoxib 40 mg either perioperatively (before operation and after operation), after operation (first dose given in the evening after surgery), or before operation (single parecoxib dose given 45 min before surgery). We measured the main outcome variables: average pain score, morphine consumption, and opioid-related symptom distress at 25, 49, and 73 h after surgery.
Perioperative parecoxib significantly (i) improved the pain score compared with both placebo and postoperative parecoxib, (ii) decreased morphine consumption, and (iii) reduced the opioid-related symptom distress score. Neither a single preoperative dose nor postoperative parecoxib (first dose given in the evening after surgery) significantly improved morphine’s analgesic effectiveness.
Perioperative parecoxib compared with postoperative parecoxib improves post-discectomy pain and results in a reduction in adverse effects associated with opioid therapy. Postoperative parecoxib, or a single pre-incisional parecoxib dose, does not significantly improve post-discectomy pain or opioid side-effects up to 3 days after surgery.
Parecoxib is the i.v. administered pro-drug of the COX-2 inhibitor valdecoxib and is available for the treatment of postoperative pain in more than 30 countries, including the European Union, Australia, New Zealand, and South Africa.
The use of COX-2 inhibitors has come under critical review due to concerns regarding their cardiovascular safety profile1 and several drugs in the class, including parecoxib, have been withdrawn from the markets of a number of countries. However, where COX-2 drugs continue to be used, questions remain regarding their optimal use in the treatment of postoperative pain. Although it has been shown that perioperative administration of COX-2 inhibitors decreases pain scores and morphine consumption compared with both placebo2–5 and postoperative COX-2 inhibitor treatment,6,7 the usefulness of pre-incisional COX inhibition has been questioned.8,9 In addition, it is not clear if the magnitude of parecoxib’s analgesic effect is clinically relevant in a multimodal postoperative pain treatment regimen that includes opioids. A clinically significant improvement of postoperative pain should also be associated with improvement in relevant outcome measures other than pain scores or morphine consumption, for example, administration of parecoxib should be shown to reduce the side-effects of analgesic therapy and interference of pain in the patient’s life. Finally, it is unclear if a single pre-incisional parecoxib dose would have relevant analgesic effects in the postoperative period.
Therefore, the aim of this study was to compare the effect of (i) perioperative, (ii) postoperative, and (iii) preoperative parcecoxib on analgesic effectiveness, that is, (A) pain intensity, (B) morphine consumption, (C) opioid-related symptom distress (SDS score),10 and the interference of pain with patient’s life [brief pain inventory (BPI) score].11 We hypothesized that perioperative parecoxib treatment would improve analgesic effectiveness compared with both placebo and postoperative parecoxib treatment, which begins in the evening after surgery.
Methods
Patients
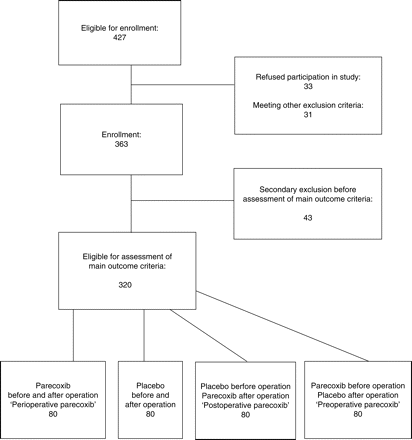
This study was approved by the local ethics committee. Four hundred and twenty-seven adult patients with painful disc herniation who were undergoing discectomy were considered for inclusion in this prospective, randomized, double-blind, placebo-controlled single centre trial (Fig. 1). Of these, 64 patients were excluded from participation for any of the following reasons: mental or physical inability to handle a patient-controlled analgesia (PCA) device or to answer pain questionnaires, ASA risk classification greater than III, preoperative opioid therapy >1 week, administration of steroids or NSAIDs within 24 h before skin incision, renal insufficiency (serum creatinine concentration >1.5 mg dl−1), allergy against sulphonamides or NSAIDs, severe liver dysfunction, congestive heart failure, history of myocardial infarction, stroke, pulmonary embolism, or gastrointestinal bleeding, patients’ refusal of study participation, or pregnancy/lactation period. Three hundred and sixty-three patients (age: 18–88 yr) gave their informed written consent to participate in the study. Of these, 43 patients were excluded before assessment of the primary criteria for the following reasons: cancellation of surgery after the first dose of the study medication, administration of NSAIDs or opioids other than morphine, patients’ desire to be excluded from the study, and patients’ discharge before assessment of main criteria. All 320 patients eligible for assessment of the main outcome criteria received a PCA device (Multifuse, B. Braun, Melsungen, Germany) upon arrival in the post-anaesthesia care unit (PACU).
Measurements
At 25, 49, and 73 h after surgery, patients recorded in a pre-printed diary their average pain level during the previous 24 h. Patients rated their distress from opioid-related symptoms in terms of frequency (rarely, occasionally, frequently, and almost constantly), severity (slight, moderate, severe, and very severe), and degree of bother (a little bit, somewhat, quite a bit, and very much). Symptoms included fatigue, drowsiness, inability to concentrate, confusion, dizziness, constipation, itching, difficulty with urination, nausea, and retching/vomiting. Furthermore, the modified BPI-short form (mBPI-sf) severity and impairment score were obtained.
Systolic and diastolic arterial pressures were measured twice daily, and serious adverse events were monitored by daily chart review and by interviewing the neurosurgeon in charge for this study (P.D.K.). We aimed at monitoring for perioperative myocardial infarction, stroke, pulmonary embolism, deep vein thrombosis, or gastrointestinal bleeding.
Interventions
On the day before scheduled surgery, analgesic medication was restricted to dipyrone (metamizol, Novalgin©, Aventis Pharma, Frankfurt, Germany) 500 mg orally upon request and given no later than 12:00 p.m. The first dose of study drug was administered i.v. 45 min before surgery by the anaesthetist in charge. Subsequent doses were administered after discharge from the recovery room by nursing staff in charge of the neurosurgical unit at 12, 24, 36, 48, 60, and 72 h after surgery. Patients received a PCA device (Multifuse, B. Braun, Melsungen, Germany) upon arrival in the PACU containing a morphine solution with a concentration of 1 mg ml−1. After administration of a loading dose via the PCA pump, the device was set to administer a bolus dose of 1.5 mg with no continuous opioid infusion, with a lockout time of 10 min, and an upper limit of 20 mg morphine/4 h.
A study nurse experienced in acute pain assessment assessed variables 25, 49, and 73 h (3 h) after surgery and at least 1 h after administration of the study drug.
The type of general anaesthesia was left at the decision of the anaesthetist.
Randomization and blinding procedures
An anaesthetist not engaged in data collection performed randomization with a computer-generated random list. Patients were randomized to four treatments (Fig. 1): (1) parecoxib 40 mg twice a day throughout (‘perioperative parecoxib’, n=80), (2) placebo before operation, parecoxib 40 mg 12 h after skin closure, and subsequently parecoxib 40 mg twice a day (‘postoperative parecoxib’, n=80), (3) parecoxib 40 mg before operation and placebo subsequently (‘preoperative parecoxib’, n=80), or (4) placebo throughout (n=80).
Statistical analysis
Average pain score, morphine consumption, and the opioid-related SDS 25 h after surgery were defined as primary outcome variables of analgesic effectiveness. Differences in values of variables between treatment groups were evaluated using the Mann–Whitney U-test. Using a hierarchical sequence, variables of analgesic effectiveness could be tested with an alpha-error of 5%.12 Within this approach, after a non-significant result, further testing is considered only for exploratory intention.
Sample size estimation was based on data on the effect of rofecoxib after spine surgery,13 and arthroscopic knee surgery13 (‘preventive’ analgesia data, variables morphine consumption and pain score), and on data on the effect of parecoxib in abdominal surgery (variable ‘composite opioid-related SDS score’).14 We expected a difference between perioperative parecoxib and placebo in pain score of 0.4 (sd 0.5), in morphine consumption of 10 mg (sd 12 mg), and in SDS of 0.2 (sd 0.3). Between perioperative parecoxib and postoperative parecoxib, we expected a difference in pain score of 0.3 (sd 0.5), in morphine consumption of 8 mg (sd 12 mg), and in SDS score of 0.1 (sd 0.2). On the basis of these assumptions, we calculated that a number of 80 patients per group would provide an 80% power to detect differences in the main outcome variables between perioperative parecoxib and both placebo and postoperative treatment (P=0.05).
Descriptive statistics for continuous variables are reported as means and standard deviation unless stated otherwise. Data were analysed using SPSS software, version 12.0 (Chicago, IL, USA).
Results
Perioperative parecoxib
Compared with placebo
Twenty-five hours after surgery, patients treated with perioperative parecoxib reported lower average pain scores and lower opioid-related symptom distress scores, and consumed less morphine (see Table 1 and Fig. 2). Furthermore, BPI impairment and severity score were lower in the ‘perioperative parecoxib’ group (Table 1).
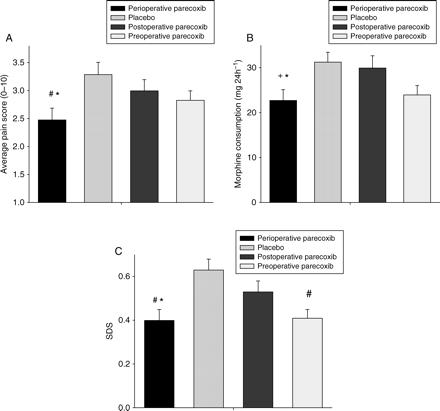
Main variables of analgesic effectiveness 25 h after discectomy. (a) Average pain score, (b) morphine consumption, and (c) composite opioid-related SDS. Patients treated with ‘perioperative parecoxib’ had significantly lower pain scores and SDS scores, and required less morphine compared with both placebo and postoperative parecoxib treatment. Means (sem) of 320 patients. #P<0.005 vs placebo, *P<0.05 vs postoperative parecoxib, +P<0.01 vs placebo.
Analgesic effects and side-effects 25 h after skin closure. Data from 320 patients after spine surgery. Means (sd). *Eleven-point numerical rating scale, 0=minimum intensity, 10=maximum intensity. †P<0.005 vs ‘perioperative parecoxib’; ‡P<0.05 vs ‘perioperative parecoxib; ¶P<0.01 vs ‘perioperative parecoxib’; §P<0.005 vs placebo; ‖P<0.001 vs ‘perioperative parecoxib’; #P<0.05 vs placebo. SDS, symptom distress score; BPI, brief pain inventory
| . | Perioperative parecoxib . | Placebo . | Postoperative parecoxib . | Preoperative parecoxib . |
|---|---|---|---|---|
| Average pain score* | 2.48 (1.84) | 3.29 (1.96)† | 3.00 (1.77)‡ | 2.83 (1.52) |
| Morphine consumption (mg) | 22.8 (19.2) | 31.3 (21.8)¶ | 30.1 (23.6)‡ | 24.9 (18.6) |
| Opiate SDS | 0.40 (1.04) | 0.63 (0.5)† | 0.53 (0.44)‡ | 0.41 (0.38)§ |
| BPI impairment score* | 1.26 (1.5) | 2.27 (1.84)‖ | 1.77 (1.31)# | 1.54 (1.09)‡ |
| BPI severity score* | 2.90 (1.81) | 3.78 (1.9)† | 3.40 (1.59)‡ | 3.31 (1.48)# |
| . | Perioperative parecoxib . | Placebo . | Postoperative parecoxib . | Preoperative parecoxib . |
|---|---|---|---|---|
| Average pain score* | 2.48 (1.84) | 3.29 (1.96)† | 3.00 (1.77)‡ | 2.83 (1.52) |
| Morphine consumption (mg) | 22.8 (19.2) | 31.3 (21.8)¶ | 30.1 (23.6)‡ | 24.9 (18.6) |
| Opiate SDS | 0.40 (1.04) | 0.63 (0.5)† | 0.53 (0.44)‡ | 0.41 (0.38)§ |
| BPI impairment score* | 1.26 (1.5) | 2.27 (1.84)‖ | 1.77 (1.31)# | 1.54 (1.09)‡ |
| BPI severity score* | 2.90 (1.81) | 3.78 (1.9)† | 3.40 (1.59)‡ | 3.31 (1.48)# |
Analgesic effects and side-effects 25 h after skin closure. Data from 320 patients after spine surgery. Means (sd). *Eleven-point numerical rating scale, 0=minimum intensity, 10=maximum intensity. †P<0.005 vs ‘perioperative parecoxib’; ‡P<0.05 vs ‘perioperative parecoxib; ¶P<0.01 vs ‘perioperative parecoxib’; §P<0.005 vs placebo; ‖P<0.001 vs ‘perioperative parecoxib’; #P<0.05 vs placebo. SDS, symptom distress score; BPI, brief pain inventory
| . | Perioperative parecoxib . | Placebo . | Postoperative parecoxib . | Preoperative parecoxib . |
|---|---|---|---|---|
| Average pain score* | 2.48 (1.84) | 3.29 (1.96)† | 3.00 (1.77)‡ | 2.83 (1.52) |
| Morphine consumption (mg) | 22.8 (19.2) | 31.3 (21.8)¶ | 30.1 (23.6)‡ | 24.9 (18.6) |
| Opiate SDS | 0.40 (1.04) | 0.63 (0.5)† | 0.53 (0.44)‡ | 0.41 (0.38)§ |
| BPI impairment score* | 1.26 (1.5) | 2.27 (1.84)‖ | 1.77 (1.31)# | 1.54 (1.09)‡ |
| BPI severity score* | 2.90 (1.81) | 3.78 (1.9)† | 3.40 (1.59)‡ | 3.31 (1.48)# |
| . | Perioperative parecoxib . | Placebo . | Postoperative parecoxib . | Preoperative parecoxib . |
|---|---|---|---|---|
| Average pain score* | 2.48 (1.84) | 3.29 (1.96)† | 3.00 (1.77)‡ | 2.83 (1.52) |
| Morphine consumption (mg) | 22.8 (19.2) | 31.3 (21.8)¶ | 30.1 (23.6)‡ | 24.9 (18.6) |
| Opiate SDS | 0.40 (1.04) | 0.63 (0.5)† | 0.53 (0.44)‡ | 0.41 (0.38)§ |
| BPI impairment score* | 1.26 (1.5) | 2.27 (1.84)‖ | 1.77 (1.31)# | 1.54 (1.09)‡ |
| BPI severity score* | 2.90 (1.81) | 3.78 (1.9)† | 3.40 (1.59)‡ | 3.31 (1.48)# |
At 49 and 73 h after skin closure, perioperative parecoxib compared with placebo still improved pain score, morphine consumption, composite SDS score, and BPI impairment score [49 h: 0.86 (1.01) (peri-OP) vs 1.56 (1.57) (placebo), P<0.005 and 73 h: 0.54 (0.82) vs 1.03 (1.17), P<0.05, respectively] and BPI severity score [49 h: 2.11 (1.7) vs 2.75 (1.93), P<0.05, and 73 h: 1.28 (1.44) vs 2.18 (1.72), P<0.005, respectively].
Comparator postoperative parecoxib
Twenty-five hours after skin closure, perioperative parecoxib improved pain score, morphine consumption, SDS, and BPI scores compared with postoperative parecoxib treatment (Table 1, Fig. 2). These differences were no longer significant during the follow-up period (49 and 73 h after skin closure).
Comparator preoperative parecoxib
Twenty-five hours after skin closure, analgesic effectiveness of preoperative parecoxib treatment was not significantly different from perioperative treatment.
Exploratory data analysis
Forty-nine hours after surgery, perioperative parecoxib treatment compared with preoperative parecoxib treatment improved average pain score and morphine consumption and BPI impairment and severity scores [impairment: 0.86 (1.01) (peri-OP) vs 1.18 (1.07) (pre-OP) and severity: 2.11 (sd 1.70) vs 2.73 (1.31), respectively]. Seventy-three hours after surgery, patients treated with perioperative parecoxib still experienced less pain [1.06 (1.36) (peri-OP) vs 1.86 (1.35) (pre-OP)].
Postoperative parecoxib
During the entire observation period, none of the variables of analgesic effectiveness was improved by postoperative parecoxib compared with placebo or preoperative parecoxib.
Preoperative parecoxib
Twenty-five hours after surgery, preoperative parecoxib improved the composite opioid-related SDS compared with placebo. SDS also tended to be improved (exploratory P=0.08) 49 h after surgery.
Neither systolic nor diastolic arterial pressure differed between treatment groups during the 4-day observation period. No episodes of myocardial infarction, stroke, pulmonary embolism, deep vein thrombosis, or gastrointestinal bleeding were observed during the observation period.
Discussion
At the time of assessment of the main criteria of analgesic effectiveness, multimodal pain therapy with perioperative parecoxib decreased pain score, morphine consumption, and the opioid-related SDS compared with both placebo, and postoperative parecoxib starting up in the evening after surgery. Postoperative parecoxib did not improve criteria of analgesic effectiveness at any point during the 3-day observation period.
The review of Ong and colleagues15 and the accompanying editorial by Kissin16 define pre-emptive analgesia as superiority of preoperative vs postoperative administration, and preventive analgesia as an effect persisting beyond the presence of the analgesic drug in the biophase. These beneficial effects of preoperative COX-2 inhibition can only be assumed when analgesic effectiveness of perioperative treatment is superior to both placebo and postoperative treatment.16–18 To our knowledge, this has been shown so far in a single study only.7 Reuben and colleagues reported that analgesic duration, defined as the time from completion of surgery until first opioid use, was longer and pain scores were significantly lower in those patients having received orally pre- vs postoperative rofecoxib. However, it remains unclear whether the observed analgesic effects with preoperative oral administration really reflect preventive effects16 on postoperative pain perception, or whether they rather are attributable to pharmacokinetic differences between preoperative and postoperative treatments. Since opioids administered intraoperatively can reduce enteral absorption of drugs,19,20 lower analgesic effectiveness of the postoperative rofecoxib observed by Reuben and colleagues7 could also be due to an inadequate postoperative absorption after oral administration. In our study, however, differences in absorption between the groups can be excluded because parecoxib was administered i.v.
Since there was no pharmacokinetic assessment conducted in the present study, it is not possible to directly relate drug concentrations in the biophase to the efficacy observed for the investigated parecoxib regimens. However, simulations of the expected valdecoxib plasma concentration time profiles for the different dosing regimens used in this study (data not shown) indicate that valdecoxib plasma concentrations at 49 h and later after surgery would be similar in subjects receiving the perioperative and the postoperative regimens. The similarity in active drug concentrations suggest that the superior efficacy seen with the perioperative regimen compared with the postoperative schedule is unlikely to be solely due to differences in drug concentrations and suggest that the preventive effects on pain sensitization play some role.16,21
During the 73 h observation period, postoperative parecoxib did not improve analgesic effectiveness of morphine PCA. This observation is in contrast to other reports on postoperative effects of parecoxib, showing a decrease in pain score and morphine consumption.22–27 However, our observation is in line with the results of a recent study,28 reporting no improvement of postoperative pain after discectomy with parecoxib 40 mg administered 45 min before the end of surgery. Therefore, it might be that the effect of preoperative parecoxib administration on postoperative pain is of particular importance after excision of a painful disc herniation. Prostaglandin production may already be an issue before operation in the region of the herniated disc.29 This suggestion is supported by the finding that samples obtained from herniated cervical30 and lumbar31 disc tissues show increased concentrations of PGE2. Preoperative COX-2 induction from disc herniation may potentiate perioperative sensitization evoked by surgery and postoperative inflammation.32 Accordingly, perioperative but not preoperative or postoperative COX-2 inhibition alone may optimally prevent postoperative pain. Our data along with the data of Grundmann and colleagues28 suggest that when parecoxib is considered for multimodal post-discectomy pain treatment, the first dose should be administered before operation, and treatment should be maintained after operation, to optimize analgesic effectiveness.
Although the mean improvement of pain scores with perioperative parecoxib of 0.8 points on the five-point NRS (25% change compared with placebo) may be considered small, we believe that the magnitude of the effect was relevant, because decrease in pain scores was associated with an improvement of important health outcome variables, the mBPI-sf, and the opioids SDS. Previous data on variables of analgesic efficacy other than pain scores are scarce. In a meta-analysis,33 COX-2 inhibitors improved significantly opioid-related adverse events, that is, vomiting, constipation, and pruritus, in only four of 26 studies. The authors argued that the poor reporting quality of adverse events could be one reason for the low success rate. We used the validated SDS score10 for assessment of opioid symptom distress, and found that perioperative parecoxib treatment improved opioid symptom distress (main criterion in our study) compared with postoperative parecoxib treatment, which supports the exploratory results of other studies that applied the SDS score as a secondary outcome criterion.3,6,26,34–37
In summary, our data show that timing of administration is a critical factor for a successful treatment of post-discectomy pain with parecoxib. Perioperative parecoxib compared with postoperative parecoxib improves postoperative pain and opioids SDS. In contrast, postoperative parecoxib did not improve analgesic efficacy at any point during the 73 h observation period, suggesting that parecoxib may have some preventive effects on post-discectomy pain.
Funding
Investigator initiated trial funded by Pfizer, Germany. Pfizer did not participate in generation of the study design or interpretation of results.
Acknowledgements
We are grateful to Clifford Woolf, Professor of Anaesthesia Research, Massachusetts General Hospital, Boston, for his useful comments and criticism. We are also grateful to Markus Neuhaeuser, Professor of Statistics, Koblenz University of Applied Science, Germany, for his supervision of statistical methods and Nancy Chamberlin, Beth Israel Deaconess Medical Centre, Boston, for her language revision. Pfizer did not participate in generation of the study design and interpretation of the results.





